Pain
Nursing Diagnoses:
Acute Pain/Chronic Pain/Impaired Comfort
related to the disease process, injury, or surgical procedure
Desired Outcome:
The patient's subjective report of pain using a pain scale, family's report, and behavioral and/or physiologic indicators reflect that pain is either reduced or at an acceptable level within 1-2 hr.
| ASSESSMENT/INTERVENTIONS | RATIONALES |
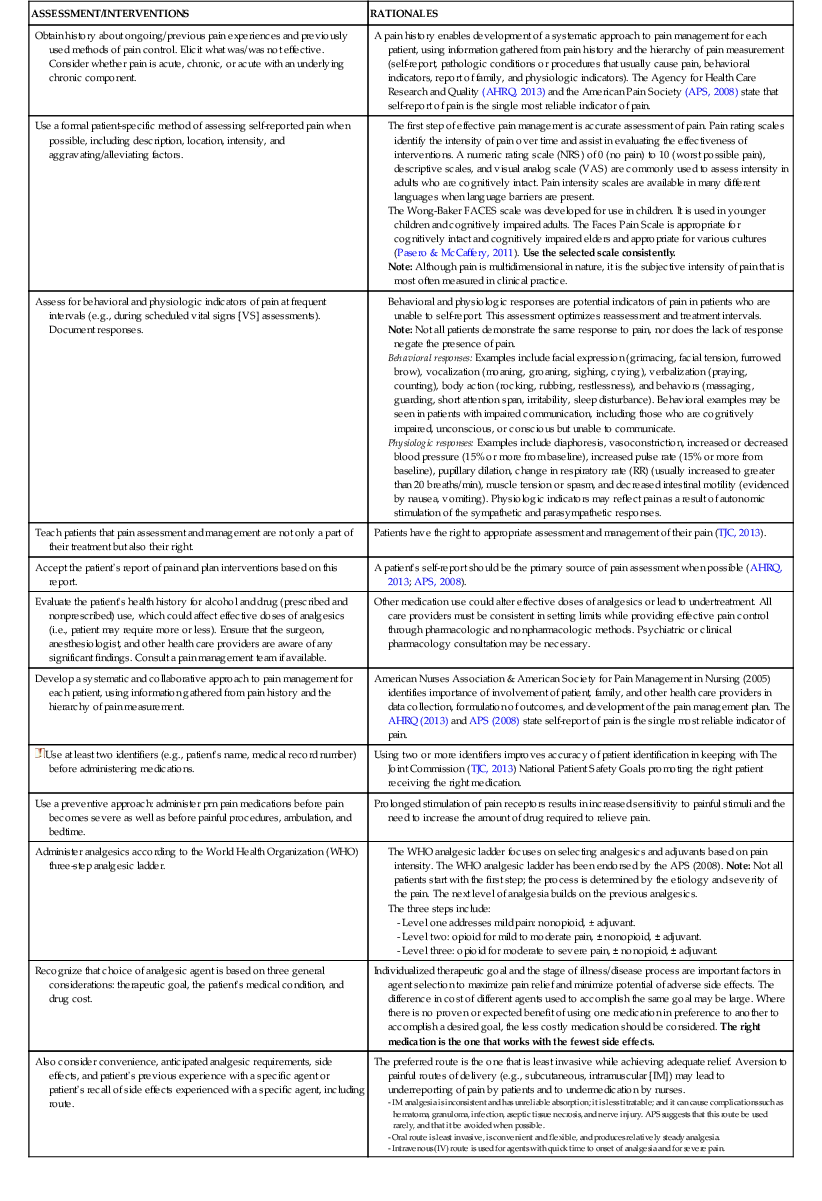
| Obtain history about ongoing/previous pain experiences and previously used methods of pain control. Elicit what was/was not effective. Consider whether pain is acute, chronic, or acute with an underlying chronic component. | A pain history enables development of a systematic approach to pain management for each patient, using information gathered from pain history and the hierarchy of pain measurement (self-report, pathologic conditions or procedures that usually cause pain, behavioral indicators, report of family, and physiologic indicators). The Agency for Health Care Research and Quality (AHRQ, 2013) and the American Pain Society (APS, 2008) state that self-report of pain is the single most reliable indicator of pain. |
| Use a formal patient-specific method of assessing self-reported pain when possible, including description, location, intensity, and aggravating/alleviating factors. | The first step of effective pain management is accurate assessment of pain. Pain rating scales identify the intensity of pain over time and assist in evaluating the effectiveness of interventions. A numeric rating scale (NRS) of 0 (no pain) to 10 (worst possible pain), descriptive scales, and visual analog scale (VAS) are commonly used to assess intensity in adults who are cognitively intact. Pain intensity scales are available in many different languages when language barriers are present. The Wong-Baker FACES scale was developed for use in children. It is used in younger children and cognitively impaired adults. The Faces Pain Scale is appropriate for cognitively intact and cognitively impaired elders and appropriate for various cultures (Pasero & McCaffery, 2011). Use the selected scale consistently. Note: Although pain is multidimensional in nature, it is the subjective intensity of pain that is most often measured in clinical practice. |
| Assess for behavioral and physiologic indicators of pain at frequent intervals (e.g., during scheduled vital signs [VS] assessments). Document responses. | Behavioral and physiologic responses are potential indicators of pain in patients who are unable to self-report. This assessment optimizes reassessment and treatment intervals. Note: Not all patients demonstrate the same response to pain, nor does the lack of response negate the presence of pain. Behavioral responses: Examples include facial expression (grimacing, facial tension, furrowed brow), vocalization (moaning, groaning, sighing, crying), verbalization (praying, counting), body action (rocking, rubbing, restlessness), and behaviors (massaging, guarding, short attention span, irritability, sleep disturbance). Behavioral examples may be seen in patients with impaired communication, including those who are cognitively impaired, unconscious, or conscious but unable to communicate. Physiologic responses: Examples include diaphoresis, vasoconstriction, increased or decreased blood pressure (15% or more from baseline), increased pulse rate (15% or more from baseline), pupillary dilation, change in respiratory rate (RR) (usually increased to greater than 20 breaths/min), muscle tension or spasm, and decreased intestinal motility (evidenced by nausea, vomiting). Physiologic indicators may reflect pain as a result of autonomic stimulation of the sympathetic and parasympathetic responses. |
| Teach patients that pain assessment and management are not only a part of their treatment but also their right. | Patients have the right to appropriate assessment and management of their pain (TJC, 2013). |
| Accept the patient's report of pain and plan interventions based on this report. | A patient's self-report should be the primary source of pain assessment when possible (AHRQ, 2013; APS, 2008). |
| Evaluate the patient's health history for alcohol and drug (prescribed and nonprescribed) use, which could affect effective doses of analgesics (i.e., patient may require more or less). Ensure that the surgeon, anesthesiologist, and other health care providers are aware of any significant findings. Consult a pain management team if available. | Other medication use could alter effective doses of analgesics or lead to undertreatment. All care providers must be consistent in setting limits while providing effective pain control through pharmacologic and nonpharmacologic methods. Psychiatric or clinical pharmacology consultation may be necessary. |
| Develop a systematic and collaborative approach to pain management for each patient, using information gathered from pain history and the hierarchy of pain measurement. | American Nurses Association & American Society for Pain Management in Nursing (2005) identifies importance of involvement of patient, family, and other health care providers in data collection, formulation of outcomes, and development of the pain management plan. The AHRQ (2013) and APS (2008) state self-report of pain is the single most reliable indicator of pain. |
| Using two or more identifiers improves accuracy of patient identification in keeping with The Joint Commission (TJC, 2013) National Patient Safety Goals promoting the right patient receiving the right medication. | |
| Use a preventive approach: administer prn pain medications before pain becomes severe as well as before painful procedures, ambulation, and bedtime. | Prolonged stimulation of pain receptors results in increased sensitivity to painful stimuli and the need to increase the amount of drug required to relieve pain. |
| Administer analgesics according to the World Health Organization (WHO) three-step analgesic ladder. | The WHO analgesic ladder focuses on selecting analgesics and adjuvants based on pain intensity. The WHO analgesic ladder has been endorsed by the APS (2008). Note: Not all patients start with the first step; the process is determined by the etiology and severity of the pain. The next level of analgesia builds on the previous analgesics. - Level one addresses mild pain: nonopioid, ± adjuvant. - Level two: opioid for mild to moderate pain, ± nonopioid, ± adjuvant. - Level three: opioid for moderate to severe pain, ± nonopioid, ± adjuvant. |
| Recognize that choice of analgesic agent is based on three general considerations: therapeutic goal, the patient's medical condition, and drug cost. | Individualized therapeutic goal and the stage of illness/disease process are important factors in agent selection to maximize pain relief and minimize potential of adverse side effects. The difference in cost of different agents used to accomplish the same goal may be large. Where there is no proven or expected benefit of using one medication in preference to another to accomplish a desired goal, the less costly medication should be considered. The right medication is the one that works with the fewest side effects. |
| Also consider convenience, anticipated analgesic requirements, side effects, and patient's previous experience with a specific agent or patient's recall of side effects experienced with a specific agent, including route. | The preferred route is the one that is least invasive while achieving adequate relief. Aversion to painful routes of delivery (e.g., subcutaneous, intramuscular [IM]) may lead to underreporting of pain by patients and to undermedication by nurses. - IM analgesia is inconsistent and has unreliable absorption; it is less titratable; and it can cause complications such as hematoma, granuloma, infection, aseptic tissue necrosis, and nerve injury. APS suggests that this route be used rarely, and that it be avoided when possible. - Oral route is least invasive, is convenient and flexible, and produces relatively steady analgesia. - Intravenous (IV) route is used for agents with quick time to onset of analgesia and for severe pain. |
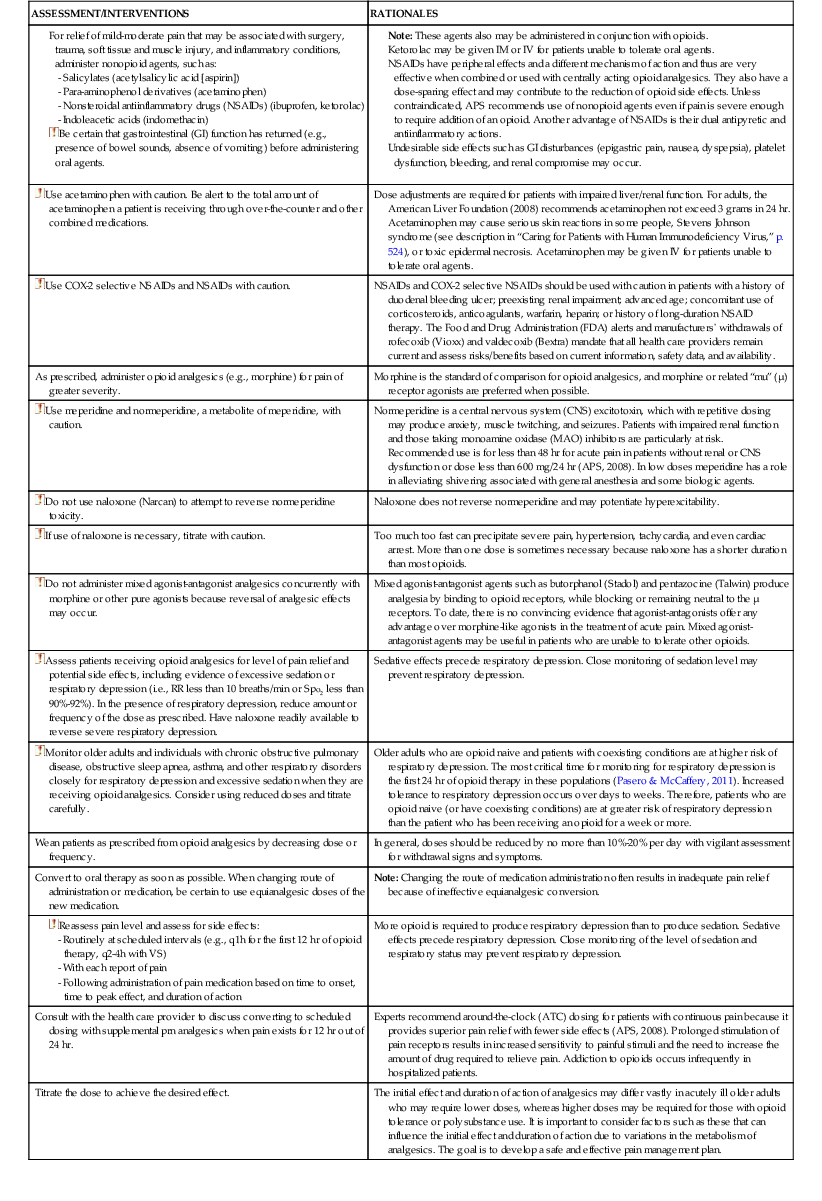
For relief of mild-moderate pain that may be associated with surgery, trauma, soft tissue and muscle injury, and inflammatory conditions, administer nonopioid agents, such as: - Salicylates (acetylsalicylic acid [aspirin]) - Para-aminophenol derivatives (acetaminophen) - Nonsteroidal antiinflammatory drugs (NSAIDs) (ibuprofen, ketorolac) - Indoleacetic acids (indomethacin)
| Note: These agents also may be administered in conjunction with opioids. Ketorolac may be given IM or IV for patients unable to tolerate oral agents. NSAIDs have peripheral effects and a different mechanism of action and thus are very effective when combined or used with centrally acting opioid analgesics. They also have a dose-sparing effect and may contribute to the reduction of opioid side effects. Unless contraindicated, APS recommends use of nonopioid agents even if pain is severe enough to require addition of an opioid. Another advantage of NSAIDs is their dual antipyretic and antiinflammatory actions. Undesirable side effects such as GI disturbances (epigastric pain, nausea, dyspepsia), platelet dysfunction, bleeding, and renal compromise may occur. |
| Dose adjustments are required for patients with impaired liver/renal function. For adults, the American Liver Foundation (2008) recommends acetaminophen not exceed 3 grams in 24 hr. Acetaminophen may cause serious skin reactions in some people, Stevens Johnson syndrome (see description in “Caring for Patients with Human Immunodeficiency Virus,” p. 524), or toxic epidermal necrosis. Acetaminophen may be given IV for patients unable to tolerate oral agents. | |
| NSAIDs and COX-2 selective NSAIDs should be used with caution in patients with a history of duodenal bleeding ulcer; preexisting renal impairment; advanced age; concomitant use of corticosteroids, anticoagulants, warfarin, heparin; or history of long-duration NSAID therapy. The Food and Drug Administration (FDA) alerts and manufacturers' withdrawals of rofecoxib (Vioxx) and valdecoxib (Bextra) mandate that all health care providers remain current and assess risks/benefits based on current information, safety data, and availability. | |
| As prescribed, administer opioid analgesics (e.g., morphine) for pain of greater severity. | Morphine is the standard of comparison for opioid analgesics, and morphine or related “mu” (µ) receptor agonists are preferred when possible. |
| Normeperidine is a central nervous system (CNS) excitotoxin, which with repetitive dosing may produce anxiety, muscle twitching, and seizures. Patients with impaired renal function and those taking monoamine oxidase (MAO) inhibitors are particularly at risk. Recommended use is for less than 48 hr for acute pain in patients without renal or CNS dysfunction or dose less than 600 mg/24 hr (APS, 2008). In low doses meperidine has a role in alleviating shivering associated with general anesthesia and some biologic agents. | |
| Naloxone does not reverse normeperidine and may potentiate hyperexcitability. | |
| Too much too fast can precipitate severe pain, hypertension, tachycardia, and even cardiac arrest. More than one dose is sometimes necessary because naloxone has a shorter duration than most opioids. | |
| Mixed agonist-antagonist agents such as butorphanol (Stadol) and pentazocine (Talwin) produce analgesia by binding to opioid receptors, while blocking or remaining neutral to the µ receptors. To date, there is no convincing evidence that agonist-antagonists offer any advantage over morphine-like agonists in the treatment of acute pain. Mixed agonist-antagonist agents may be useful in patients who are unable to tolerate other opioids. | |
| Sedative effects precede respiratory depression. Close monitoring of sedation level may prevent respiratory depression. | |
| Older adults who are opioid naive and patients with coexisting conditions are at higher risk of respiratory depression. The most critical time for monitoring for respiratory depression is the first 24 hr of opioid therapy in these populations (Pasero & McCaffery, 2011). Increased tolerance to respiratory depression occurs over days to weeks. Therefore, patients who are opioid naive (or have coexisting conditions) are at greater risk of respiratory depression than the patient who has been receiving an opioid for a week or more. | |
| Wean patients as prescribed from opioid analgesics by decreasing dose or frequency. | In general, doses should be reduced by no more than 10%-20% per day with vigilant assessment for withdrawal signs and symptoms. |
| Convert to oral therapy as soon as possible. When changing route of administration or medication, be certain to use equianalgesic doses of the new medication. | Note: Changing the route of medication administration often results in inadequate pain relief because of ineffective equianalgesic conversion. |
| More opioid is required to produce respiratory depression than to produce sedation. Sedative effects precede respiratory depression. Close monitoring of the level of sedation and respiratory status may prevent respiratory depression. | |
| Consult with the health care provider to discuss converting to scheduled dosing with supplemental prn analgesics when pain exists for 12 hr out of 24 hr. | Experts recommend around-the-clock (ATC) dosing for patients with continuous pain because it provides superior pain relief with fewer side effects (APS, 2008). Prolonged stimulation of pain receptors results in increased sensitivity to painful stimuli and the need to increase the amount of drug required to relieve pain. Addiction to opioids occurs infrequently in hospitalized patients. |
| Titrate the dose to achieve the desired effect. | The initial effect and duration of action of analgesics may differ vastly in acutely ill older adults who may require lower doses, whereas higher doses may be required for those with opioid tolerance or polysubstance use. It is important to consider factors such as these that can influence the initial effect and duration of action due to variations in the metabolism of analgesics. The goal is to develop a safe and effective pain management plan. |
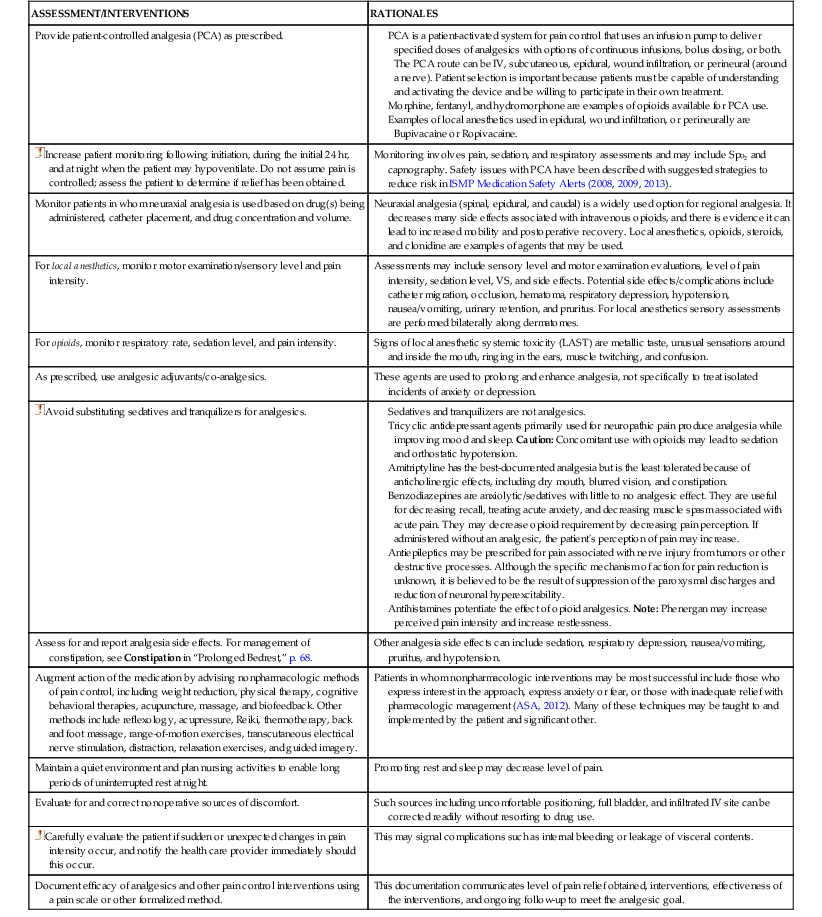
| Provide patient-controlled analgesia (PCA) as prescribed. | PCA is a patient-activated system for pain control that uses an infusion pump to deliver specified doses of analgesics with options of continuous infusions, bolus dosing, or both. The PCA route can be IV, subcutaneous, epidural, wound infiltration, or perineural (around a nerve). Patient selection is important because patients must be capable of understanding and activating the device and be willing to participate in their own treatment. Morphine, fentanyl, and hydromorphone are examples of opioids available for PCA use. Examples of local anesthetics used in epidural, wound infiltration, or perineurally are Bupivacaine or Ropivacaine. |
| Monitoring involves pain, sedation, and respiratory assessments and may include Spo2 and capnography. Safety issues with PCA have been described with suggested strategies to reduce risk in ISMP Medication Safety Alerts (2008, 2009, 2013). | |
| Monitor patients in whom neuraxial analgesia is used based on drug(s) being administered, catheter placement, and drug concentration and volume. | Neuraxial analgesia (spinal, epidural, and caudal) is a widely used option for regional analgesia. It decreases many side effects associated with intravenous opioids, and there is evidence it can lead to increased mobility and postoperative recovery. Local anesthetics, opioids, steroids, and clonidine are examples of agents that may be used. |
| For local anesthetics, monitor motor examination/sensory level and pain intensity. | Assessments may include sensory level and motor examination evaluations, level of pain intensity, sedation level, VS, and side effects. Potential side effects/complications include catheter migration, occlusion, hematoma, respiratory depression, hypotension, nausea/vomiting, urinary retention, and pruritus. For local anesthetics sensory assessments are performed bilaterally along dermatomes. |
| For opioids, monitor respiratory rate, sedation level, and pain intensity. | Signs of local anesthetic systemic toxicity (LAST) are metallic taste, unusual sensations around and inside the mouth, ringing in the ears, muscle twitching, and confusion. |
| As prescribed, use analgesic adjuvants/co-analgesics. | These agents are used to prolong and enhance analgesia, not specifically to treat isolated incidents of anxiety or depression. |
Sedatives and tranquilizers are not analgesics. Tricyclic antidepressant agents primarily used for neuropathic pain produce analgesia while improving mood and sleep. Caution: Concomitant use with opioids may lead to sedation and orthostatic hypotension. Amitriptyline has the best-documented analgesia but is the least tolerated because of anticholinergic effects, including dry mouth, blurred vision, and constipation. Benzodiazepines are anxiolytic/sedatives with little to no analgesic effect. They are useful for decreasing recall, treating acute anxiety, and decreasing muscle spasm associated with acute pain. They may decrease opioid requirement by decreasing pain perception. If administered without an analgesic, the patient's perception of pain may increase. Antiepileptics may be prescribed for pain associated with nerve injury from tumors or other destructive processes. Although the specific mechanism of action for pain reduction is unknown, it is believed to be the result of suppression of the paroxysmal discharges and reduction of neuronal hyperexcitability. Antihistamines potentiate the effect of opioid analgesics. Note: Phenergan may increase perceived pain intensity and increase restlessness. | |
| Assess for and report analgesia side effects. For management of constipation, see Constipation in “Prolonged Bedrest,” p. 68. | Other analgesia side effects can include sedation, respiratory depression, nausea/vomiting, pruritus, and hypotension. |
| Augment action of the medication by advising nonpharmacologic methods of pain control, including weight reduction, physical therapy, cognitive behavioral therapies, acupuncture, massage, and biofeedback. Other methods include reflexology, acupressure, Reiki, thermotherapy, back and foot massage, range-of-motion exercises, transcutaneous electrical nerve stimulation, distraction, relaxation exercises, and guided imagery. | Patients in whom nonpharmacologic interventions may be most successful include those who express interest in the approach, express anxiety or fear, or those with inadequate relief with pharmacologic management (ASA, 2012). Many of these techniques may be taught to and implemented by the patient and significant other. |
| Maintain a quiet environment and plan nursing activities to enable long periods of uninterrupted rest at night. | Promoting rest and sleep may decrease level of pain. |
| Evaluate for and correct nonoperative sources of discomfort. | Such sources including uncomfortable positioning, full bladder, and infiltrated IV site can be corrected readily without resorting to drug use. |
| This may signal complications such as internal bleeding or leakage of visceral contents. | |
| Document efficacy of analgesics and other pain control interventions using a pain scale or other formalized method. | This documentation communicates level of pain relief obtained, interventions, effectiveness of the interventions, and ongoing follow-up to meet the analgesic goal. |