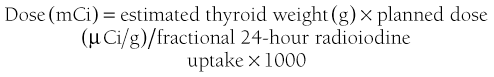Treatment of Graves’ Disease
Etiologic treatment of Graves’ disease is not available at present. Therefore, the major aim of current methods is correction of thyrotoxicosis in all cases and treatment of Graves’ ophthalmopathy when present and severe (detailed elsewhere in this textbook). Additional goals include relief of compressive symptoms from large goiters.
Correction of thyroid hormone overproduction can be obtained by inhibition of its synthesis or release or by ablation of thyroid tissue via surgery or radioiodine. In addition, the peripheral metabolism of thyroid hormone can be altered in a favorable way by available drugs.
Selection of the treatment option from among those available should be made after careful consideration of the many variables inherent in any given patient, but there is room for the patient's choice in most cases. The patient should therefore participate in the choice of treatment, after thorough information on the therapeutic alternatives.
Overview of Therapeutic Tools in Graves’ Disease Thyrotoxicosis
Clinical Pharmacology: Thionamides (methimazole, carbimazole, and propylthiouracil) were first described and introduced into clinical practice in the early 1940s.9 The major action of thionamides is to inhibit the organification of iodine and coupling of iodotyrosines, thus blocking the synthesis of thyroid hormones.237,283 Carbimazole is not active as it is, but it is almost completely converted to methimazole in the body, and their effects are comparable. Propylthiouracil has the additional effect of partially inhibiting the conversion of T4 to T3 in peripheral tissues, but this effect is of limited clinical value. Methimazole is at least 10 times more potent than propylthiouracil. The pharmacologic properties of the two major thionamides are compared in Table 9-5.
Table 9-5
Pharmacologic Properties of Thionamides: Comparison Between Methimazole and Propylthiouracil
| Property | Methimazole | Propylthiouracil |
| Relative potency | >10 (up to 50) | 1 |
| Administration route | PO | PO |
| Absorption | Almost complete | Almost complete |
| Binding to serum proteins | Negligible | 75% |
| Serum half-life (hours) | 4-6 | 1-2 |
| Duration of action (hours) | >24 | 12-24 |
| Transplacental passage | Low | Lower |
| Levels in breast milk | Low | Lower |
| Inhibition of deiodinase | No | Yes |
Both methimazole and propylthiouracil are very effective in controlling hyperthyroidism, and their side-effect record is quite similar, thus making the choice between the two drugs largely a matter of personal preference and local availability. Antithyroid drugs do not block the release of preformed thyroid hormones, so euthyroidism is not obtained until intrathyroidal hormone and iodine stores are depleted. This process requires 1 to 6 weeks, depending on factors such as disease activity, initial levels of circulating thyroid hormones, and intrathyroidal hormone and iodine stores. Large goiters with abundant deposits of thyroid hormone, especially when iodine excess is present, often show a delayed response to thionamides.
The main problem with thionamide treatment is the high relapse rate of thyrotoxicosis after discontinuation of even long-term treatment. Although remission rates on the order of 50% to 60% within 1 year after withdrawal of thionamides have been reported in a few series,284 in most studies hyperthyroidism recurred in 50% to 80% of patients, depending on the duration of the follow-up period.285-288 Remission rates have been decreasing in the last decades, possibly as a result of increased iodine supply in the diet.289,290 A practical problem is that no single test or combination of tests will accurately separate patients who will relapse from those who will not. Size of the goiter before or during antithyroid drug treatment, HLA-DR3 typing, TRAb or TPOAb levels, serum thyroglobulin concentrations, thyroid echogenicity by ultrasound, circulating activated T cells, T-cell subset ratios, and presence of Graves’ ophthalmopathy, have all been indicated as significant pretreatment risk factors for relapse, but none has the required sensitivity or specificity to predict the outcome in individual patients. The presence of a large goiter seems to be the most significant predictor of future hyperthyroidism relapse287 (Fig. 9-8). Similar considerations apply during treatment to parameters such as T3 suppression of 99mTc uptake, the thyrotropin-releasing hormone test, and the T3/T4 ratio at the time of discontinuation of thionamide therapy.286,287,291-297 In children, a longer initial duration of euthyroid state with antithyroid drugs seems to be the only variable related to the risk of relapse.288 Regardless of age, a good predictor of relapse of hyperthyroidism is a positive TSAb test before discontinuation of medical treatment. However, even when TSAbs disappear, the chances of relapse are still high, ranging from 20% to 50%.
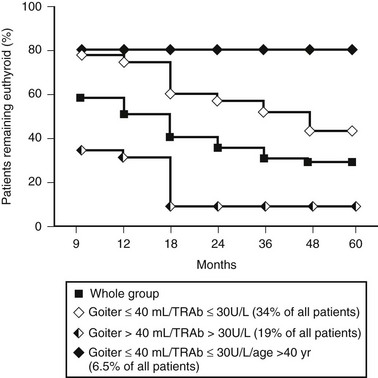
FIGURE 9-8 Effect of goiter size and thyroid-stimulating hormone receptor antibody (TRAb) level at the time of antithyroid drug discontinuation on the incidence of relapses of hyperthyroidism in a cohort of patients with Graves’ disease. (Modified from Vitti P, Rago T, Chiovato L et al: Clinical features of patients with Graves’ disease undergoing remission after antithyroid drug treatment. Thyroid 7:369–375, 1997.)
Most relapses of hyperthyroidism occur within 3 to 6 months after medical therapy is discontinued, and more than two thirds of patients who relapse will do so within 2 years. However, hyperthyroidism can also recur much later. Late evolution to primary hypothyroidism can be observed as well, mainly in patients who remain euthyroid after discontinuation of therapy.298,299 Relapse of hyperthyroidism after a full cycle of thionamides is a strong indication for alternative treatments such as radioiodine or thyroidectomy, but a second course of the drug can be given, for example, to adolescents, while bearing in mind that people who have relapsed once are more likely to do so after a second cycle.
Minor side effects of thionamides have been reported in 1% to 15% of patients, but the average appears to be 6%. Pruritus, skin rash, and much less commonly, urticaria are the most prominent manifestations.283 Arthralgias have also been reported. These side effects frequently resolve spontaneously despite continued therapy. However, when any of them occur, it is generally advisable to replace one thionamide with the other, although cross-sensitivity to these drugs may occur. Antihistamine drugs can be used to control mild side effects. Slight elevations in liver enzyme levels have often been reported, and sometimes it is difficult to distinguish this effect of thionamides from the effect of thyrotoxicosis itself. When detected, serious alterations in liver function test results must be monitored closely because toxic hepatitis may develop suddenly.
Serious side effects are uncommon with thionamides and are observed in approximately 3 of every 1000 patients.283,300 Agranulocytosis (granulocyte count <500/mm3) may be observed with both methimazole and propylthiouracil. Agranulocytosis has been reported more frequently in elderly patients, but it can occur at any age. It is most often detected within the first 3 to 4 months after starting therapy. Agranulocytosis may develop so suddenly that even weekly white blood cell counts may not detect it. Agranulocytosis is typically initially manifested by fever and evidence of infections, most often in the upper respiratory tract. Instructing all patients taking antithyroid drugs to report these symptoms immediately is probably the safest measure for immediate detection of this complication. Routine white blood cell counts should be performed in all patients before initiation of treatment, because mild leukopenia is common in Graves’ disease and needs to be distinguished from a drug reaction. In addition to prompt discontinuation of the antithyroid drug, treatment of agranulocytosis includes the administration of broad-spectrum antibiotics and growth factors to stimulate bone marrow. Patients usually recover within 2 to 3 weeks, but some deaths have been reported from this complication.
Cholestatic (mostly observed with methimazole) or necrotic (mostly observed with propylthiouracil) hepatitis is another rare but severe complication of thionamide treatment, being associated with significant mortality and sometimes requiring liver transplantation.283,300 Vasculitis and lupus-like syndromes are even rarer. In the presence of a major adverse reaction to thionamides, such as agranulocytosis, hepatitis, or vasculitis, prompt withdrawal of the drug is mandatory. The risk of cross-reactivity is such that switching from methimazole to propylthiouracil or vice versa is not recommended when side effects are severe, so alternative treatments of thyrotoxicosis must be sought.
Treatment Strategies: The purpose of treatment of Graves’ disease hyperthyroidism with antithyroid drugs is to achieve stable euthyroidism. Antithyroid drugs can be used either as preparatory treatment before surgery or radioiodine or as a primary management tool of the disease in an attempt to induce long-term remission of thyrotoxicosis. A direct effect of methimazole and propylthiouracil on the immune system has been proposed to explain the observation that a minority of patients experience long-lasting remissions of Graves’ disease thyrotoxicosis after withdrawal of these drugs.148 This view is suggested by the following lines of evidence. In some follow-up studies, patients treated with antithyroid drugs had a higher remission rate than did those to whom β-blockers alone were administered,148 but randomized studies have never been performed. Treatment of Graves’ disease with antithyroid drugs is followed by a fall in the levels of circulating TRAb, AbTg, and AbTPO,146,301-303 although this effect is not dose dependent.284 In vitro experiments have suggested a down-regulating effect of methimazole on antigen presentation,304 and in vivo studies have shown that the drug is able to reduce the severity of experimental thyroiditis.305,306 Despite these observations, the immunosuppressive effect of thionamides remains controversial. A decrease in circulating thyroid antibody titer has also been observed in hyperthyroid patients treated with perchlorate, a drug with different pharmacologic properties.307 Restoration of euthyroidism per se might be responsible for the decrease in thyroid autoantibodies, through a direct effect of thyroid hormone on the immune system.308 The natural history of the disease, which like that of many other autoimmune disorders is characterized by cycles of spontaneous relapse and remission, could also explain the reduction in thyroid autoantibody titers. In other words, a course of 12 to 24 months of thionamides would merely be a way of keeping the patient euthyroid while waiting for the autoimmune process to subside or even vanish.
Thionamide treatment is usually started with high doses (20 to 40 mg/day of methimazole or 200 to 400 mg/day of propylthiouracil). Doses of methimazole above 40 mg/day are rarely necessary. When long-term thionamide treatment is planned, one of two treatment strategies is currently used:
1. Maintenance of euthyroidism with the minimum effective dose throughout the trial period, with thyroid function tests performed every 1 to 3 months. The minimum dose capable of maintaining euthyroidism is derived by “back-titration” every 4 to 6 weeks.
2. Administration of fixed, relatively high doses of thionamide in combination with levothyroxine to prevent iatrogenic hypothyroidism, the so-called block-and-replace regimen.
With both schemes, patients should be kept completely euthyroid, with serum TSH levels within the normal range.
The second protocol was proposed because of the supposed immunosuppressive effect of higher doses of thionamides and because of studies suggesting a greater remission rate of hyperthyroidism in Graves’ patients treated with high doses of thionamides (60 versus 15 mg of methimazole per day).309 The addition of levothyroxine supposedly provides an extra advantage, and very high remission rates were reported in Japanese patients treated with methimazole for 6 months and then given a combination of methimazole and levothyroxine for an additional year, followed by levothyroxine alone for 3 years.309,310 However, the latter results have not been reproduced by a number of subsequent studies.301-315 In a prospective randomized trial of low (10 mg/day) versus moderately high (40 mg/day) doses of methimazole, no advantages were observed in terms of a decrease in TRAb titer or the rate of relapse of hyperthyroidism.284 The rate of adverse reactions was greater in the group of patients receiving methimazole at 40 mg/day. Thus at present, the block-and-replace regimen has no proven advantage, although one point in favor of it is that it probably requires less testing. The block-and-replacement strategy can also be useful in rare patients who experience changes from hyperthyroidism to hypothyroidism and vice versa after minimal changes in the dosage of antithyroid drugs (“brittle hyperthyroidism”). In these unusual patients, maintenance of euthyroidism is difficult with antithyroid drugs alone.
Regardless of the chosen regimen, treatment is maintained for 12 to 24 months, after which thionamide therapy is usually discontinued. Indefinite treatment even with low doses of thionamides is not a common practice.
In summary, thionamide treatment of Graves’ disease thyrotoxicosis has the major advantages of not causing permanent hypothyroidism and of limiting exposure to radiation. It is, however, associated with a very high failure rate, and in many cases it is only a way to delay thyroid ablation by radioiodine or surgery.
Iodine and Iodine-Containing Compounds
Inorganic iodine given in pharmacologic doses (as Lugol's solution or as saturated solution of potassium iodide [SSKI]) decreases its own transport into the thyroid, inhibits iodine organification (the Wolff-Chaikoff effect), and blocks the release of T4 and T3 from the gland.316 As an additional advantage, iodine sharply decreases the vascularity of the thyroid in Graves’ disease.317,318 These effects are, however, transient and last a few days or weeks, after which the antithyroid action of pharmacologic iodine is lost and thyrotoxicosis recurs or may worsen. Therefore, iodine therapy is now used only for short periods in the preparation of patients for surgery, after euthyroidism has already been achieved and maintained with thionamides. Iodine is also used in the management of severe thyrotoxicosis (thyroid storm) because of its ability to inhibit thyroid hormone release acutely. The usual dose of Lugol's solution is 3 to 5 drops three times a day, and that of SSKI is 1 to 3 drops three times daily.
Oral cholecystographic agents (iopanoic acid and sodium ipodate) produce a very rapid fall in the serum concentration of thyroid hormones.319-321 These agents act through a dual mechanism: virtually complete inhibition of the peripheral conversion of T4 to T3 and prevention of thyroid hormone secretion because of the inorganic iodine released from the drug.322,323 The first action is the predominant one and makes these drugs highly effective when rapid management of thyrotoxicosis is needed. The rate of fall in T3 levels after treatment is started approaches the physiologic half-life of the hormone, approximately 1 day. Although early reports suggested that these iodinated compounds could be successfully used as a primary therapy for hyperthyroidism in doses of 0.5 to 1 g/day, they proved to be of limited value in long-term treatment because of the escape of thyroid hormone synthesis from the blocking effect of iodine.324-326 Moreover, they provide the thyroid with a load of iodine, which may make the use of radioiodine unfeasible for some weeks. Therefore, these agents are ideally used in emergency situations when rapid control of thyrotoxicosis is needed, in preparation for thyroid surgery, or while waiting for the effect of radioiodine therapy. In the latter case, they may also be used to prevent or correct the transient thyrotoxicosis caused by the release of preformed thyroid hormone, as it can occur after radioiodine treatment.
Perchlorate
Perchlorate inhibits active transport of iodine into the thyroid.327 Side effects (gastric irritation) and adverse effects (aplastic anemia) are not infrequent and preclude the use of perchlorate in the long-term management of Graves’ disease thyrotoxicosis.328 In conjunction with thionamides, perchlorate has been successfully used as a tool for depleting the thyroidal iodine overload in amiodarone-induced hyperthyroidism.329
Lithium
It has been reported that the use of lithium may be beneficial in Graves’ patients undergoing radioiodine therapy.330 If given on the day of thionamide withdrawal (5 days before radioiodine) for 19 days, lithium has been found to reduce markedly the extent of thyrotoxicosis either due to thionamide withdrawal before radioiodine or to radioiodine itself after its administration. The dosage used was 900 mg/day, but even doses of 450 mg/day seem to be effective. The effects of lithium possibly reflect a direct inhibitory action on hormone release or on intrathyroidal iodine turnover. In the patients treated with this type of therapy, no adverse effects have been reported, even though psychic effects are in theory possible.331
β-Adrenergic Antagonist Drugs
Many of the manifestations of thyrotoxicosis, especially those in the cardiovascular system, are due to hyperactivity or hypersensitivity of the sympathetic nervous system. Blockade of β-adrenergic receptors thus ameliorates the manifestations of thyrotoxicosis that are related to sympathetic action, such as tachycardia, palpitation, tremor, and anxiety.332 This effect is much faster than that obtained with thionamides, and for this reason β-blockers are important in the early management of thyrotoxicosis. β-Adrenergic antagonists do not affect thyroid hormone synthesis and release or their action at the level of many tissues, such as bone. These drugs should not be used alone in Graves’ disease thyrotoxicosis, except for short periods before and/or after radioiodine therapy. Since the introduction of propranolol, a number of new agents became available with a longer duration (atenolol, metoprolol, and nadolol) or with greater cardioselectivity (atenolol, metoprolol, bisoprolol). None of these drugs seems to have an advantage over the others, and the choice largely depends on the personal experience of the physician. Propranolol has the additional advantage of mild inhibition of the peripheral conversion of T4 to T3,333 but the real clinical advantage provided by this pharmacologic property is unclear. The usual contraindications to β-adrenergic antagonists, such as asthma, should be taken into account. β-Blocker use can be rapidly tapered and discontinued once stable euthyroidism is obtained with thionamides, radioiodine, or surgery.
Glucocorticoids
Glucocorticoids in high doses inhibit the peripheral conversion of T4 to T3. In Graves’ disease thyrotoxicosis, glucocorticoids appear to decrease T4 secretion by the thyroid, possibly by immune suppression, but the efficiency and duration of this effect are unknown. Because of the significant side effects associated with the long-term use of glucocorticoids and the effectiveness of alternative treatments, use of these drugs in the management of Graves’ hyperthyroidism is not justified. On the contrary, the immunosuppressive effect of glucocorticoids in high doses is commonly exploited in the treatment of ophthalmopathy and dermopathy of Graves’ disease. In severe thyrotoxicosis or thyroid storm, short-term glucocorticoid administration may be used as a general supportive treatment.
Radioiodine
Radioactive isotopes of iodine were initially used in the treatment of Graves’ disease in the 1940s.10 Among different radioactive isotopes, 131I is the agent of choice in the treatment of thyroid hyperfunction because of its half-life and its favorable emission profile. After oral administration, radioiodine is completely absorbed, rapidly concentrated, oxidized, and organified by thyroid follicular cells—exactly the same fate of 127I (the stable isotope). Thyroid cells are destroyed by the ionizing effects of β particles with an average path length of 1 to 2 mm. One microcurie of 131I retained per gram of thyroid tissue delivers approximately 70 to 90 rad. The early biologic effects of radioiodine include necrosis of follicular cells and vascular occlusion, which fully develop over a period of weeks to months after a single dose of radioiodine. As a consequence, control of hyperthyroidism requires at least weeks or months to be achieved. Long-term effects include shorter survival, impaired replication of surviving cells with atrophy and fibrosis, and a chronic inflammatory response resembling Hashimoto's thyroiditis. These later effects account for the development of hypothyroidism even years after treatment.334
Treatment Strategies: In Graves’ disease, the goal of radioiodine therapy is to destroy enough thyroid tissue to cure thyrotoxicosis with one dose of 131I, possibly given in a single session. Three outcomes of radioiodine treatment are possible:
1. The patient is rendered stably euthyroid. Achievement of euthyroidism was once considered the “success” situation, but it might not be the ideal outcome in patients with coexistent Graves’ ophthalmopathy, in whom greater thyroid antigen ablation may be desirable.
2. The patient remains thyrotoxic. This result is, of course, a failure and requires a second treatment.
3. The patient becomes permanently hypothyroid. This outcome is now considered an acceptable consequence of radioiodine treatment, because correction of hypothyroidism with levothyroxine is easy, safe, and inexpensive.
In any patient with Graves’ disease who receives radioiodine, the likelihood of these possible outcomes depends on the amount of radioiodine that is delivered and retained by the thyroid tissue and on other incompletely understood individual factors. The latter make it impossible to predict a successful dose in every single patient. The dose of radioiodine to be administered is most often calculated on the basis of thyroid size and uptake of 131I and is determined by using the following equation335:
The planned dose varies according to the aim of treatment and ranges from 80 to 200 µCi/g in different centers. In some centers, standard fixed doses are given. Lower doses result in a lower rate of early (within 1 year) hypothyroidism, but at the expense of a higher rate of recurrent or persistent thyrotoxicosis and thus the necessity of a second or, less frequently, a third dose. Even patients given a lower dose and remaining euthyroid in the first year have a high incidence of late-onset hypothyroidism. The cumulative incidence of post-radioiodine hypothyroidism steadily increases at a rate of 2% to 3% new cases per year. The overall incidence of post-radioiodine hypothyroidism approaches a total of 40% at 5 years and 60% or more at 10 years.336 Therefore, in many centers, including our own, the strategy of radioiodine treatment is to give a dose of radioiodine that ensures cure in the highest number of patients while being aware that most of the “cured” patients will eventually become hypothyroid. Hypothyroidism should be regarded as a common outcome of radioiodine treatment rather than a true complication, and it can be easily and economically controlled with levothyroxine substitution treatment. Further arguments can be made in favor of this approach. Recurrence of thyrotoxicosis can rarely occur, even in patients who were euthyroid after the first dose of radioiodine. These recurrences are psychologically disturbing for the patient and may carry additional cardiovascular risk, especially in the elderly. Moreover, in some centers, treatment of moderate to severe Graves’ ophthalmopathy, when needed, is delayed until permanent correction of thyrotoxicosis is achieved, so rapid attainment of this goal is desirable.
With the use of relatively high delivered doses of 131I, 150 to 200 µCi/g of estimated thyroid weight, nearly 70% of patients are cured after one dose of 131I, 25% require a second dose, and rare patients need a third or fourth dose. Large goiter size, rapid iodine turnover, and adjunctive therapy with antithyroid drugs too soon after radioiodine are associated with a higher rate of persistence of hyperthyroidism, but other individual factors are also likely to exist. The decision to give a second dose of 131I is not usually made before 6 to 12 months after the first one, when firm demonstration of persistence of thyrotoxicosis can be obtained. Transient hypothyroidism may be observed in the first 6 months after 131I therapy. To correctly detect these cases, levothyroxine substitution should be initiated at submaximal doses so that TSH can be rechecked 2 to 4 months later; if high, the hypothyroidism is very likely to be permanent.
Short-Term Adverse Effects of Radioiodine: Transient exacerbation of mild to moderate preexisting Graves’ ophthalmopathy may occur in the first few months after radioiodine therapy,337 although this experience is not shared by all investigators, and the effect may be due to untreated hypothyroidism, according to others.338 Because worsening of ophthalmopathy is transient and effectively controlled with a short course of oral corticosteroids, the presence of mild to moderate ophthalmopathy is not a contraindication to the use of radioiodine.337 When severe Graves’ ophthalmopathy is present, specific treatment with high-dose oral or intravenous corticosteroids and/or external radiation therapy should be started soon after radioiodine treatment.
Radioiodine treatment causes a radiation-induced acute thyroiditis that can rarely be clinically manifested 3 or 4 days after administration of the isotope by pain and swelling in the neck. This side effect is benign and self-limited and can be treated with a short course of antiinflammatory drugs. The destruction of thyroid tissue after radioiodine treatment also induces the release of preformed thyroid hormone from the gland, which can result in reexacerbation of thyrotoxicosis in the weeks after the procedure. To prevent this phenomenon by depleting intrathyroidal stores of hormones, a few months’ course of thionamide is often given and discontinued 3 to 8 days before radioiodine administration. True relapses of hyperthyroidism after thionamide withdrawal in preparation for 131I administration may also occur and account for the increase in thyroid hormone levels observed after radioiodine administration.339 Because the effect of radioiodine is relatively delayed, several months may be required for complete control of the thyrotoxicosis. While waiting for the effect of radioiodine, a short course of antithyroid drugs can be initiated 2 weeks after treatment and the dose smoothly tapered in the following months. Earlier thionamide treatment has been associated with a higher rate of radioiodine failure. Alternatively, iopanoic acid or sodium ipodate can be administered a few days after the administration of radioiodine. This form of treatment has the advantage of rapidly controlling both thyrotoxicosis from radiation thyroiditis and transient relapse of hyperthyroidism. As mentioned earlier, lithium is another valid alternative.
Potential Long-Term Risks of Radioiodine: Radioisotope treatment of a benign disorder such as Graves’ disease may raise concern regarding possible carcinogenic effects and the risk of genetic damage (i.e., the risk of causing germline mutations in the offspring of patients treated during the child-bearing years). Although external head and neck irradiation is undoubtedly associated with an increased rate of thyroid carcinoma,340,341 no association between radioiodine treatment for hyperthyroidism and thyroid cancer was found in large epidemiologic studies.336 Similarly, no evidence has indicated that radioiodine therapy for hyperthyroidism increases the patient's risk for leukemia or solid tumors.336 A minimal increase in the risk of gastric cancer 10 years or more after treatment was found in a survey in Sweden342 but not in large epidemiologic studies in the United Kingdom336 and the United States.343 No association between radioiodine treatment of hyperthyroidism and congenital abnormalities in subsequent offspring has been observed.344 In addition, no evidence of chromosome damage was found in one recent study in children and adolescents given radioactive iodine for thyroid carcinoma.345 A rough estimate of the dose to the ovaries is about 0.2 rad/mCi of administered 131I. Thus the dose to the ovaries in a patient receiving 10 mCi of radioiodine is similar to that received from a barium enema or intravenous pyelography. It has been calculated that if a genetic risk induced by 131I really does exist, the risk would be only 0.003% per rad of parental gonadal exposure and therefore a very small fraction of the spontaneous incidence of genetic disorders.346 Increased risks of all-cause and circulatory deaths versus age- and period-specific mortality were reported but found to be associated with mild hypothyroidism prior to thyroxine therapy rather than to radioiodine treatment per se.347 The experience accumulated in more than 50 years of radioiodine treatment of hyperthyroidism has shown that potential long-term risks are absent or negligible in the adult population. Unfortunately, no large studies are available on such risks in the pediatric population. Data from populations exposed to radioactive isotopes after the Chernobyl accident indicate that in infancy the thyroid is much more susceptible to radioiodine-induced carcinogenesis.348,349 These observations are based on data that are very skewed with regard to the amount and duration of exposure to the radioactive fallout and therefore cannot be extrapolated to the therapeutic use of radioiodine. The Cooperative Thyrotoxicosis Therapy Follow-up Study showed that thyroid cancer develops in children treated with low but not with high doses of 131I.350 Therefore, it has been recently suggested to treat children with doses higher than those given to adults for Grave's hyperthyroidism.351 Nevertheless, because of concerns and uncertainties especially for the risk of cancer of other organs, there is still no consensus for recommending radioiodine treatment for persons younger than 16 to 18 years.
Surgery
The aim of surgical treatment in Graves’ hyperthyroidism is to reduce the excessive secretion of thyroid hormones and prevent relapse of thyrotoxicosis by removal of enough thyroid tissue. Subtotal thyroidectomy has for a long time been the choice of surgery for Graves’ disease. The classic procedure consists of removing the bulk of the gland, with a few grams of tissue left in both lobes. With subtotal thyroidectomy, many patients remain euthyroid but are exposed to the risk of future relapse of thyrotoxicosis. A significant number of patients treated with subtotal thyroidectomy also become hypothyroid352 (Fig. 9-9). Therefore, lifelong surveillance is needed after subtotal thyroidectomy. Near-total thyroidectomy consists of the removal of most thyroid tissue, with only subcentimeter fragments left in sensitive regions, such as around the laryngeal recurrent nerve or the parathyroid glands. Near-total thyroidectomy has more recently been performed in patients with Graves’ disease and results in a higher rate of hypothyroidism but a much smaller incidence of recurrent hyperthyroidism.352,353 Because of the very low risk of relapse of thyrotoxicosis, near-total thyroidectomy has become the preferred operation in specialized centers.352 Total thyroidectomy (e.g., removal of all visible thyroid tissue) may have the additional advantage of removing virtually all thyroidal autoantigens, and thus it might have a positive influence on the course of Graves’ ophthalmopathy when present.354
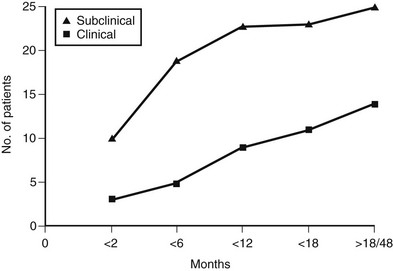
FIGURE 9-9 Prevalence of subclinical and clinical hypothyroidism in a cohort of patients with Graves’ disease treated by subtotal thyroidectomy. (Modified from Miccoli P, Vitti P, Rago T et al: Surgical treatment of Graves’ disease: subtotal or total thyroidectomy? Surgery 120:1020–1025, 1996.)
Preparation of the patient for thyroid surgery is of paramount importance. A course of thionamide treatment is recommended to restore and maintain euthyroidism and to deplete intrathyroidal stores of hormones that could be released during surgery. The preoperative administration (10 days) of inorganic iodine induces involution of the gland and a reduction in vascularity.317 Another approach has been proposed, namely, preparation with propranolol and iodine alone, which allows earlier surgery. In the absence of a real need for rapid surgery, however, this approach should be discouraged because it exposes patients to unnecessary risks. When emergency surgery is needed, oral cholecystographic agents represent the fastest way to obtain euthyroidism.
Besides the common nonspecific complications of surgery and anesthesia, thyroid surgery exposes patients to certain specific complications, including thyroid storm (which is now extremely rare), bleeding, injury to the recurrent laryngeal nerve, and hypoparathyroidism. In particular, the risk of laryngeal nerve injury and hypoparathyroidism cannot be disregarded. The incidence of these complications depends on the skill and experience of the surgeon and may range from 2% in specialized centers with wide experience in thyroid surgery up to 10% to 15% in some series.354-359
As stated earlier, these two potential complications need to be carefully explained to the patient when the discussion of treatment options is started. Postoperative thyroid function largely depends on the extent of thyroidectomy and duration of follow-up. Insufficient tissue removal results in persistent hyperthyroidism or in later relapse of hyperthyroidism, which may occur in 5% to 10% of patients within 5 years and in up to 40% within 30 years after limited thyroidectomy. Recurrence of hyperthyroidism is particularly undesirable, because a second operation is technically more difficult than the first one and involves a higher risk of complications. With few exceptions, therefore, such patients should be treated with radioiodine. Extensive thyroidectomy results in postoperative thyroid failure, which always occurs after total and near-total thyroidectomy and often after subtotal thyroidectomy. In the first year after surgery, hypothyroidism has been reported in percentages of patients ranging from 5% to 60%.352 Late-onset cases develop in an additional 1% to 3% per year. Hypothyroidism is easily and economically treated with levothyroxine replacement therapy, so thyroid failure after thyroidectomy for Graves’ hyperthyroidism should not be considered a real complication.
Making The Choice of Treatment
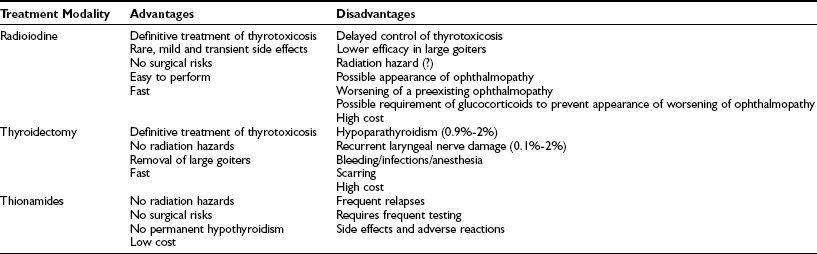
In the preceding paragraphs, the possible therapeutic tools for Graves’ disease thyrotoxicosis (antithyroid drugs, radioiodine, and surgery) have been examined. Some of them represent alternatives to the others. It is therefore important to discuss the relevant variables that will guide both the patient and the clinician in the choice of the best option for each particular case. General advantages and disadvantages of three major tools for the treatment of Graves’ disease hyperthyroidism are listed in Table 9-6. Scientific evidence collected over the past 50 years or so is of invaluable help in making the correct decision. In many cases, the choice is in fact guided by preference, by personal experience of both the patient and the physician, or by environmental conditions (for example, the lack of an experienced surgeon or a well-equipped nuclear medicine facility). A survey of practice in Europe showed that a large majority of European endocrinologists still preferred medical treatment with thionamides in many cases of Graves’ hyperthyroidism, including the most typical one, a woman in her forties with a medium-sized goiter. Ninety-five percent of European endocrinologists would have chosen this approach in a younger patient (e.g., a 19-year-old woman).360 In contrast, when the same questionnaire was given to North American endocrinologists, 69% selected radioiodine for the older patient, and 27% chose it for the 19-year-old hyperthyroid female.361 In both groups, thyroidectomy was not considered adequate in small to medium-sized goiters. Such wide differences in the perception of the best option for the treatment of Graves’ disease not only reflect different traditions and experiences worldwide but also the fact that none of the available options has a clear-cut advantage over the others.
Table 9-6
Advantages and Disadvantages of Available Treatment Modalities for Graves’ Disease Thyrotoxicosis
In a typical uncomplicated Graves’ disease case, for example, a middle-aged woman with a medium-sized goiter and mild Graves’ ophthalmopathy, it is reasonable to offer the patient a trial of antithyroid drugs in an attempt to obtain persistent remission of hyperthyroidism, with the knowledge that this result will be obtained in a small, albeit significant minority of cases. At the same time, the patient can also be presented with the possibility of definitive treatment of hyperthyroidism with radioiodine and should receive an explanation regarding the fact that the most likely result will be permanent hypothyroidism. In many cases, however, this basic approach to the treatment of hyperthyroidism must be modified in light of other factors, which makes the choice of treatment somewhat less optional.
Age
Although radioiodine has been effectively used in adolescents and young adults with no adverse effects,362,363 because of the lack of studies on the long-term effects of radioiodine, we usually exert caution in this situation, and our primary choice for treatment is antithyroid drugs, at least until the patient is 20 years old or so. Thyroidectomy may also be advised in children and adolescents who are allergic or noncompliant with antithyroid drugs. However, subtotal thyroidectomy may be more hazardous in children, in whom acute complications are reported in 16% to 35% and permanent complications in up to 8%.364,365
In women in the reproductive age, pregnancy must be delayed for at least 4 months after radioiodine administration and possibly for 1 year.366 Therefore, the treatment plan should be designed with the patient according to her family plans, with antithyroid drug treatment being relatively safe and effective during pregnancy (see later heading).
Opposite considerations can be made in elderly subjects, in whom faster definitive correction of hyperthyroidism may be warranted. Relapse of hyperthyroidism after antithyroid drug treatment increases the cardiovascular risk in elderly patients, and surgery may present excessive risks in these patients. Therefore, radioiodine can be considered the best choice in the elderly.
Goiter Size and Associated Nodular Thyroid Disease
Large goiters are relatively resistant to 131I and often require multiple treatments before correction of hyperthyroidism. Moreover, radioiodine only induces partial and slow shrinkage of the goiter. Therefore, when the patient has a large goiter, especially if compressive symptoms are present, surgery is the best choice of treatment, provided that an experienced surgeon is available.
Surgery is also recommended when Graves’ disease is superimposed on endemic goiters with multiple cold nodules, which are not expected to respond with shrinkage to radioiodine. Finally, surgery is mandatory when a suspicion of malignancy cannot be ruled out in an associated single cold nodule, regardless of the size of the goiter.
Graves’ Ophthalmopathy
The possible untoward effects of radioiodine on preexisting mild or moderate Graves’ ophthalmopathy have been considered previously. The presence of severe Graves’ ophthalmopathy requiring active treatment may modify the clinical approach. In a recent survey, most European thyroidologists chose antithyroid drug treatment in an index case with severe ophthalmopathy,367 which implies that most of them were concerned with a possible worsening of Graves’ ophthalmopathy after radioiodine treatment. The fact that thionamide treatment does not appear to be associated with worsening of ophthalmopathy was also responsible for this choice. As an alternative option, prompt definitive treatment of thyrotoxicosis by radioiodine or surgery may be warranted in the hope that the ongoing autoimmune process in the thyroid gland may drive the one in the orbit and that removal of possible cross-reacting thyroidal antigen(s) may improve orbital autoimmunity. In this regard, evidence in patients with differentiated thyroid carcinoma and coexistent thyroid autoimmunity indicates that successful removal of the thyroid by total thyroidectomy and radioiodine ablation is followed by the disappearance of autoantibodies.368 Indeed, it has been recently reported a more beneficial outcome to glucocorticoid treatment in patients with Graves’ ophthalmopathy treated by total thyroid ablation (near-total thyroidectomy followed by radioiodine ablation) compared with patients treated with thyroidectomy not followed by radioiodine.354
Relapses of thyrotoxicosis after discontinuation of treatment with antithyroid drugs are also frequent and may result in exacerbation of the orbital inflammatory process. Therefore, in these cases we usually advise rapid ablation of the thyroid with radioiodine or surgery, closely followed by the appropriate treatment of Graves’ ophthalmopathy.
Concurrent Nonthyroidal Illnesses
The presence of nonthyroidal illnesses, especially heart disease, requires special attention in the choice of treatment of Graves’ thyrotoxicosis. In these patients, surgery may be contraindicated or involve excessive risk, and relapses of thyrotoxicosis can worsen the concurrent heart disease. For these reasons, radioiodine is the therapeutic tool of choice, preceded by accurate short-term antithyroid drug preparation and followed by protection from post-radioiodine thyrotoxicosis by thionamides, iopanoic acid (see following discussion), or lithium. A similar approach can be taken with other nonthyroidal disorders that can be affected by thyroid function status, such as diabetes and severe psychiatric diseases, although in the latter conditions thyroidectomy can be advised as well.
Patient's Choice and Environmental Factors
From the preceding, it is apparent that only rarely is the choice of one treatment modality mandatory. As a consequence, the patient can often be directly involved in the decision-making process after complete information on advantages and disadvantages. For example, some patients may have a disproportionate perception and fear of the meaning of “treatment with radioactive compounds.” In other cases, the presence of a goiter is perceived as disfiguring, and the patient may require surgery for cosmetic purposes. Yet other patients may be reluctant to undergo long-term drug therapy or may wish to solve the problem quickly. Other considerations that must be taken into account when selecting the treatment modality are the availability of an experienced surgeon and/or an experienced nuclear medicine facility. In some countries, restrictive legislation makes administration of sufficient doses of radioiodine more difficult.
Special Situations
A higher incidence of abortion, preterm delivery, low-birth-weight infants, and neonatal mortality is seen in pregnancies complicated by maternal hyperthyroidism.369 Besides fetal complications, hyperthyroidism may also cause maternal complications such as heart failure, eclampsia, and thyroid storm during delivery. Recognition of thyrotoxicosis during gestation or before a planned pregnancy warrants immediate and appropriate treatment inasmuch as pregnancies in which hyperthyroidism is fully controlled have excellent outcomes in mothers with Graves’ disease.369 It should also be added that for this reason pregnancy is not contraindicated in patients with Graves’ disease, and conversely, thyrotoxicosis is not a reason for recommending abortion.
When Graves’ disease is diagnosed in a woman planning pregnancy, pregnancy can be allowed after restoration of euthyroidism with thionamide drugs and treatment continued during pregnancy (see later). Alternatively, the radioiodine option can be offered because of a lack of evidence of an association between 131I treatment of hyperthyroidism and congenital abnormalities in subsequent offspring. Current guidelines recommend that pregnancy be delayed for at least 4 months after radioiodine therapy,369 but in a conservative approach, we usually advise the patient to wait for 1 year, when thyroid function is fully normalized and the outcome of treatment is clear.
Thyroidectomy may also be considered as an alternative because of more rapid restoration of euthyroidism.
Radioiodine therapy is absolutely contraindicated during pregnancy because it may result in congenital hypothyroidism and may cause malformations. Surgery is restricted to exceptional cases.
Thionamides are the first choice for treatment in a pregnant woman with Graves’ disease. Clinical improvement with thionamides occurs after the first week, and euthyroidism may be reached after 2 to 4 weeks of therapy. Both propylthiouracil and methimazole have been used in pregnancy and are equally effective in the management of hyperthyroidism in this setting.237,369,370 In the United States, propylthiouracil has traditionally been preferred to methimazole because of methimazole's purported increased passage across the placenta and breast epithelium, and because of a reported association between methimazole use and aplasia cutis (see following). However, methimazole or its precursor, carbimazole, are used widely throughout the world to treat pregnant women. In the only in vivo human study to formally examine the possible placental transfer of propylthiouracil and methimazole, pregnant women ingested 35S-labeled compounds 2 hours before the elective termination of pregnancies in gestational age of 8 to 20 weeks.371 The ratio of fetal serum or cord blood drug levels to maternal blood levels was substantially higher for methimazole or carbimazole (0.72 to 1) than for propylthiouracil (0.27 to 0.35). The differences were attributed to the known disparity in drug binding to albumin (propylthiouracil > methimazole) and in lipid solubility, as well as to possible differences in maternal/fetal volumes of distribution, excretion, and metabolism. More recently, a study using isolated perfused human placentas found no difference in the rate or extent of transplacental passage between propylthiouracil and methimazole.372 Although it is uncertain whether this in vitro model is completely representative of in vivo events, a lack of a difference in placental transfer of propylthiouracil and methimazole is consistent with clinical observations showing similar fetal outcomes with either drug, in terms of thyroid function and congenital anomalies, and with data showing that cord blood propylthiouracil levels were similar to or higher than simultaneously obtained maternal serum propylthiouracil levels.372
Aplasia cutis is a congenital localized absence of skin that occurs spontaneously in approximately 1 in 2000 births.373 Some cases of aplasia cutis (and also of other malformations) have been reported in the offspring of mothers who had taken methimazole during pregnancy, suggesting a possible association between methimazole and aplasia cutis.237,369,370 Nevertheless, there is no definitive proof that methimazole is actually responsible for the condition, and considering the extreme rarity of this association, the fear of aplasia cutis should not prevent the use of methimazole during pregnancy, especially in those countries where propylthiouracil is not promptly available. Additional congenital malformations in newborns exposed to methimazole during the first trimester of pregnancy are choanal and esophageal atresia, minor facial abnormalities, and psychomotor delay, which either isolated or associated in rare cases with aplasia cutis define a condition named methimazole embryopathy.237,369,370 As for isolated aplasia cutis, there is no proof that methimazole is responsible for these malformations, which therefore should not prevent the use of the drug during pregnancy. In a hyperthyroid pregnant women, the first priority should always be the control of hyperthyroidism, regardless of the drug. Nevertheless, because there are no case reports of aplasia cutis in association with propylthiouracil, when promptly available, propylthiouracil might still be preferred by some physicians.
Treatment with thionamides should be monitored so that maternal FT4 is kept in the high normal range. This level will ensure fetal euthyroidism inasmuch as FT4 levels in the mother's serum are correlated with fetal FT4 levels, as assessed in cord blood.374 A low TSH level is not a reliable index to judge the adequacy of treatment, because it may not reflect changes in thyroid function as promptly as FT4 does, but it is important to test it because a high level always indicates insufficient treatment and should prompt adjustment of the thionamide dose. Because of the immunosuppressive effect of pregnancy, partial and transient remission of Graves’ disease may occur in the second and third trimesters and allow a reduction and even discontinuation of thionamide treatment (Fig. 9-10). On the other hand, relapse of hyperthyroidism is frequent in the postpartum period.99
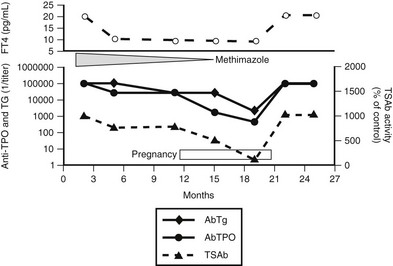
FIGURE 9-10 Course of hyperthyroidism in a patient with Graves’ disease before, during, and after pregnancy. The immunosuppressive effect of pregnancy is shown by the reduction in serum antithyroid peroxidase (TPOAb) and antithyroglobulin (TgAb) titers and by the disappearance of serum antithyroid-stimulating hormone receptor antibodies (TSAb) during the third trimester of pregnancy, which allowed discontinuation of methimazole (MMI) treatment (lower panel). Serum free thyroxine (FT4) remained within the normal range without treatment (upper panel), until a relapse of the autoimmune process (shown by an increase in the titer of the three antibodies) caused a relapse of thyrotoxicosis shortly after the end of pregnancy.
The block-and-replace regimen is not recommended in pregnancy, because much more thionamide than levothyroxine will cross the placenta and cause fetal hypothyroidism. Iodine is avoided because of the risk of fetal hypothyroidism and goiter caused by the greater sensitivity of the fetal thyroid to the Wolff-Chaikoff effect. The use of β-blockers is controversial in pregnancy, but most authorities will not recommend them, at least for prolonged periods.
Both methimazole and propylthiouracil are secreted in breast milk (methimazole more than propylthiouracil) in small amounts.237,369,370 However, maternal treatment with thionamides during lactation appears to be safe, whether it is continued after gestation or initiated in the postpartum period. For methimazole, doses of up to 20 mg daily have been documented not to affect infants’ thyroid function.375 For propylthiouracil, data available are scanty, and relatively low doses are therefore recommended. Whatever the drug, a mother should take her drug dose just after breastfeeding, which should provide a 3- to 4-hour interval before she lactates again. Although maternal hormone levels must be monitored with appropriate antithyroid drug adjustment, it appears that the child's thyroid function does not need to be checked regularly as long as somatic and mental development are progressing normally.
Surgical treatment is only occasionally indicated and may be considered in cases of poor compliance, severe drug allergy, very large goiter, associated thyroid malignancy, or the necessity of using high doses of thionamides to maintain euthyroidism. When needed, thyroidectomy is most safely performed in the second trimester. Complications of surgery, such as vocal cord paralysis or hypoparathyroidism, are disabling, and the latter may be difficult to treat during pregnancy. Levothyroxine therapy for the mother should be promptly initiated postoperatively.
Neonatal and Fetal Transfer Thyrotoxicosis
Immunoglobulins cross the placenta, and maternal TSAbs can also do so (Fig. 9-11). Because of this phenomenon, neonatal thyrotoxicosis may occur in association with maternal Graves’ disease.376 Mothers who have had Graves’ disease and are euthyroid after thyroidectomy or radioiodine treatment may still have circulating TSAbs capable of causing neonatal thyrotoxicosis. Therefore, this possibility should be considered in all pregnant women with a current or past history of Graves’ disease. Because of its pathogenesis, neonatal transfer thyrotoxicosis is always transient and spontaneously remits once maternal TSAbs disappear from the circulation, but it can cause acceleration of growth and craniosynostosis. Tachycardia, jaundice, heart failure, and failure to thrive characterize neonatal thyrotoxicosis. Onset may be delayed for a few days after delivery, until maternal thionamides clear, or rarely for a few weeks when an admixture of TBAbs and TSAbs is present in the serum. In mothers with high-titer TRAb in serum, testing for these antibodies in fetal cord blood can be performed at the time of delivery to predict the onset of neonatal thyrotoxicosis.377
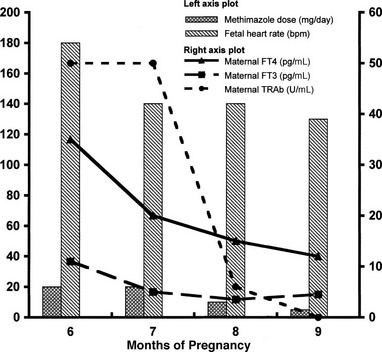
FIGURE 9-11 Fetal heart rate, maternal thyroid hormone, and maternal serum thyroid-stimulating hormone receptor antibodies (TRAb) during methimazole (MMI) treatment in a woman in whom Graves’ disease was diagnosed at the sixth month of pregnancy. FT3, Free triiodothyronine; FT4, free thyroxine.
Neonatal transfer thyrotoxicosis requires prompt treatment in close collaboration with a neonatologist. Thionamides, methimazole (0.5 to 1 mg/kg/day) or propylthiouracil (5 to 10 mg/kg/day), must be administered every 8 hours. Propranolol may be added to slow the heart rate and reduce hyperactivity. Iodine (1 drop of Lugol's solution, equivalent to 8 mg of iodine every 8 hours) is also used in addition to thionamides to inhibit the release of preformed thyroid hormones. As an alternative form of treatment, sodium ipodate alone, 0.5-g doses every 3 days, has been reported to rapidly normalize serum T4 and T3 in thyrotoxic neonates.378 In severely ill infants, glucocorticoids may be added as a general supportive measure and to block conversion of T4 to T3.
When hyperthyroidism occurs during fetal life, the diagnosis is suggested by a heart rate over 160 beats per minute after 22 weeks of gestation. The diagnosis can be confirmed by fetal cord blood sampling, but this procedure is risky, with a 1% chance of fetal loss.377 In utero treatment of hyperthyroidism may be accomplished by giving antithyroid drugs to the mother. The dose of thionamide should be adjusted to maintain a fetal heart rate of about 140 beats/min.
Graves’ Disease in Childhood and Adolescence
Graves’ disease may occur in childhood, but it is rarely seen before 10 years of age. Most cases occur around puberty, between the ages of 11 and 15. The clinical findings are often impressive, with prominent neuropsychological manifestations and an acceleration of growth that can result in premature ossification of bone end plates and reduced final height. Antithyroid drugs, radioiodine, and surgery have all been successfully used in children.363 Radioiodine has been shown to be effective in children with hyperthyroidism,362 but its long-range potential for radiation oncogenesis and gonadal damage remains to be established. Long-term follow-up studies of radioiodine therapy in children, that is, in patients who have a 60- to 70-year life expectancy, are still limited, although some authors consider this form of treatment safe.351,363 Given the above considerations, most children are treated with thionamides for long periods (3 to 4 years) in an attempt to induce stable remission or until they reach an age when radioiodine treatment or surgery is more suitable (usually 18 to 20 years of age). Long-term courses of therapy imply close medical supervision and parental involvement because compliance may be low in this age group.
Thyroidectomy is rarely performed in children and adolescents, because permanent complications occur at a relatively higher rate in this age group and have a greater impact on developmental age. Nevertheless, very large goiters or poor compliance with antithyroid drug therapy may be an indication for surgical treatment.
Hyperthyroidism in the Elderly With Heart Disease
In the elderly, hyperthyroidism and cardiac disease are often associated. Hyperthyroid symptoms may be quite different in aged patients, and symptoms related to adrenergic hyperactivity such as hyperactive reflexes, increased sweating, heat intolerance, tremor, nervousness, and increased appetite are found less frequently than in younger patients379 (Table 9-7). Weight loss is more often associated with anorexia, depression, and lethargy than with decreased appetite.380 Overall, these findings justify the expression “apathetic thyrotoxicosis.” Graves’ ophthalmopathy has been reported to be more severe in the elderly, when present.381
Hyperthyroidism may precipitate heart failure or may worsen preexisting heart conditions. Atrial fibrillation is found in 30% to 60% of patients at diagnosis.379,380 Therefore, special attention should be put into rapidly controlling hyperthyroidism, avoiding relapses, and preventing additional heart complications. When heart disease is already present at the time of diagnosis, the treatment plan should be particularly cautious and include (1) prompt restoration of euthyroidism with antithyroid drugs while independently addressing the heart disease; (2) administration of β-adrenergic antagonists; (3) definitive, ablative treatment of hyperthyroidism with radioiodine; (4) protection of the heart from the possible radioiodine-induced transient thyrotoxicosis with β-adrenergic antagonists (immediately) and/or oral cholecystographic agents (2 weeks after radioiodine) or lithium; (5) resumption of thionamide therapy 2 weeks after radioiodine administration to control hyperthyroidism while waiting for the complete effect of radioiodine; (6) strict control of thyroid function in the next 12 months; (7) cautious tapering of antithyroid drug doses until discontinuation; and (8) correction of hypothyroidism with the minimal amount of levothyroxine needed to maintain serum TSH in the normal range.
Treatment of Pretibial Myxedema
In most cases, pretibial myxedema causes little discomfort, but it can be disfiguring in others. Itching may also be a dominant symptom. Application of occlusive dressings with topical high-potency corticosteroids appears to be an effective treatment.382 Alternative treatment modalities include local injections of corticosteroids or hyaluronidase.383 The treatment needs to be repeated from time to time because the disease is characterized by a course of remissions and relapses. Eventually, stable remissions are obtained in most cases. In more severe cases, surgical excision of pseudotumorous or polypous forms has been performed with success.384,385 Systemic treatment has included plasmapheresis and high-dose corticosteroids.382 Plasmapheresis is effective, but its favorable effects are transient when used alone without long-term immunosuppression. High-dose intravenous immunoglobulins have been used as well in uncontrolled studies, but this treatment is very expensive and offers no advantages over corticosteroids.
Thyroid Storm
Thyroid storm is an acute and severe life-threatening complication of thyrotoxicosis characterized by manifestations of severe hypermetabolism with high fever, tachyarrhythmias, profuse sweating, diarrhea and vomiting, confusion, delirium, and coma.385 Congestive heart failure is often a relevant part of the picture. Thyroid storm occurs in patients with poor nutritional status and longstanding thyrotoxicosis, most often Graves’ disease, either not recognized or not adequately treated. Although the cause is thought to be an abrupt release of large quantities of stored thyroid hormones into the circulation, in many cases the mechanism cannot be found. Before the use of antithyroid drugs, thyroid surgery and radioiodine therapy were relatively common causes of thyroid storm. Infections, trauma, surgical distress, metabolic disorders, and pulmonary and cardiovascular diseases are among the other factors that may precipitate a thyroid storm. Thyroid storm used to be much more frequent in the past and was associated with very high mortality, up to 75%.386
True thyroid storm is an extremely rare event nowadays. However, less severe forms of thyrotoxic crisis are not exceptional and may be a medical emergency requiring prompt recognition and adequate treatment. Underlying nonthyroidal illnesses should be promptly recognized and specifically treated. Normalization of body temperature may require the use of cooling blankets and/or pharmacologic agents such as acetaminophen, chlorpromazine, or meperidine. Intravenous corticosteroids are beneficial in sustaining the peripheral circulation and preventing shock. Supportive measures such as oxygen and intravenous fluids should be given in case of hypoxia or dehydration. All means should be used to reduce the levels of circulating thyroid hormone. Inhibition of the synthesis and release of thyroid hormones can be achieved with the use of thionamides and iodine, but this effect is delayed and therefore not sufficient, although they are necessary. Inhibition of the peripheral conversion of T4 to T3 by iodinated contrast agents is probably the fastest way to obtain a significant reduction in circulating T3. Block of the peripheral effects of thyroid hormones by β-adrenergic antagonists (propranolol, metoprolol, atenolol) is also indicated. Removal of excess thyroid hormones from the circulation by plasmapheresis, peritoneal dialysis, extracorporeal resin perfusion, or charcoal plasma perfusion have all been reported in extreme situations. All drugs should be used in maximal doses. Patients require continuous monitoring of the electrocardiogram and an intravenous line to administer fluids and drugs. Alternatively, drugs can be delivered by nasogastric tube.
References
1. Taylor, S. Robert Graves. The Golden Years of Irish Medicine. New York: Royal Society of Medicine Services; 1989.
2. Graves, RJ. Newly observed affection of the thyroid. London Med Surg J. 1835;7:515–523.
3. Parry, CH. Collections from the Unpublished Medical Writings of the Late Caleb Hillier Parry. London: Underwood; 1825.
4. Flajani, G. Sopra un tumor freddo nell’anterior parte del collo detto broncocele. [Collezione d’osservazioni e riflessioni di chirurgia.]. 1802;3:270–273.
5. von Basedow, KA. Exophthalmos durch hypertrophie des zellgewebes in der Augenhole. Wochenschr Ges Heilk Berl. 1840;6:197.
6. Hennemannn, G. Historical aspects about the development of our knowledge of morbus Basedow. J Endocrinol Invest. 1991;14:617–624.
7. Medvei, VC. History of Clinical Endocrinology. New York: Parthenon; 1992.
8. MacKenzie, JB, MacKenzie, CG, McCollum, EV. The effect of sulfanylguanidine on the thyroid in the rat. Science. 1941;94:518–519.
9. Astwood, EB. Treatment of hyperthyroidism with thiourea and thiouracil. JAMA. 1943;122:78.
10. Sawin, CT, Becker, DV. Radioiodine and the treatment of hyperthyroidism: the early history. Thyroid. 1997;7:163–176.
11. Adams, DD, Purves, HD. Abnormal responses in the assay of thyrotropin. Proc Univ Otago Med Sch. 1956;34:11–12.
12. Nagayama Y Kaufman, KD, Seto, P, Rapoport, B. Molecular cloning, sequence and functional expression of the cDNA for the human thyrotropin receptor. Biochem Biophys Res Commun. 1989;165:1184–1190.
13. Parmentier, M, Libert, F, Maenhaut, C, et al. Molecular cloning of the thyrotropin receptor. Science. 1989;246:1620–1622.
14. Laurberg, P, Pedersen, KM, Vestergaard, H, et al. High incidence of multinodular toxic goitre in the elderly population in a low iodine intake area vs. high incidence of Graves’ disease in the young in a high iodine intake area: comparative surveys of thyrotoxicosis epidemiology in East-Jutland Denmark and Iceland. J Intern Med. 1991;229:415–420.
15. Furszyfer, J, Kurland, LT, McConahey, WM, et al. Epidemiologic aspects of Hashimoto's thyroiditis and Graves’ disease in Rochester Minnesota (1935–1967), with special reference to temporal trends. Metabolism. 1972;21:197–204.
16. Aghini-Lombardi, F, Antonangeli, L, Martino, E, et al. The spectrum of thyroid disorders in an iodine-deficient community: the Pescopagano survey. J Clin Endocrinol Metab. 1999;84:561–566.
17. Tunbridge, W, Evered, DC, Hall, R, et al. The spectrum of thyroid disease in a community: the Whickham survey. Clin Endocrinol (Oxf). 1977;7:481–493.
18. Vanderpump, MP, Tunbridge, WM, French, JM, et al. The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin Endocrinol (Oxf). 1995;43:55–68.
19. Abraham-Nordling, M, Törring, O, Lantz, M, et al. Incidence of hyperthyroidism in Stockholm, Sweden, 2003–2005. Eur J Endocrinol. 2008;158:823–827.
20. Jacobson, DL, Gange, SJ, Rose, NR, et al. Epidemiology and estimated population burden of selected autoimmune diseases in the United States. Clin Immunol Immunopathol. 1997;84:223–243.
21. Connolly, RJ, Vidor, GI, Stewart, JC. Increase in thyrotoxicosis in endemic goitre area after iodination of bread. Lancet. 1970;1:500–502.
22. Stanbury, JB, Ermans, AE, Bourdoux, P, et al. Iodine-induced hyperthyroidism: occurrence and epidemiology. Thyroid. 1998;8:83–100.
23. Burgi, H, Kohler, M, Morselli, B. Thyrotoxicosis incidence in Switzerland and benefit of improved iodine supply. Lancet. 1998;352:1034.
24. Lundgren, E, Christensen Borup, S. Decreasing incidence of thyrotoxicosis in an endemic goitre inland area of Sweden. Clin Endocrinol. 1990;33:133–138.
25. Brownlie, BE, Wells, JE. The epidemiology of thyrotoxicosis in New Zealand: incidence and geographical distribution in north Canterbury, 1983–1985. Clin Endocrinol (Oxf). 1990;33:249–259.
26. Barker, DJP, Phillips, DIW. Current incidence of thyrotoxicosis and past prevalence of goitre in 12 British towns. Lancet. 1984;2:567–570.
27. Laurberg, P, Jørgensen, T, Perrild, H, et al. The Danish investigation on iodine intake and thyroid disease, DanThyr: status and perspectives. Eur J Endocrinol. 2006;155:219–228.
28. Bülow Pedersen, I, Laurberg, P, Knudsen, N, et al. Increase in incidence of hyperthyroidism predominantly occurs in young people after iodine fortification of salt in Denmark. J Clin Endocrinol Metab. 2006;91:3830–3834.
29. Brix, TH, Kyvik, KO, Christensen, K, et al. Evidence for a major role of heredity in Graves’ disease: a population-based study of two Danish twin cohorts. J Clin Endocrinol Metab. 2001;86:930–934.
30. Ringold, DA, Nicoloff, JT, Kesler, M, et al. Further evidence for a strong genetic influence on the development of autoimmune thyroid disease: the California twin study. Thyroid. 2002;12:647–653.
31. Hall, R, Stanbury, JB. Familial studies of autoimmune thyroiditis. Clin Exp Immunol. 1967;2:719–725.
32. Hall, RO, Owen, SG, Smart, GA. Evidence for a genetic predisposition to formation of thyroid autoantibodies. Lancet. 1960;ii:187–190.
33. Jacobson, EM, Tomer, Y. The genetic basis of thyroid autoimmunity. Thyroid. 2007;17:949–961.
34. Hall, R, Dingle, PR, Roberts, DF. Thyroid antibodies: a study of first-degree relatives. Clin Genet. 1972;3:319–324.
35. Tamai, H, Ohsako, N, Takeno, K, et al. Changes in thyroid function in euthyroid subjects with family history of Graves’ disease; a follow up study of 69 patients. J Clin Endocrinol Metab. 1980;51:1123–1128.
36. Villanueva, R, Greenberg, DA, Davies, TF, et al. Sibling recurrence risk in autoimmune thyroid disease. Thyroid. 2003;13:761–764.
37. Villanueva, R, Inzerillo, AM, Tomer, Y, et al. Limited genetic susceptibility to severe Graves’ ophthalmopathy: no role for CTLA-4 but evidence for an environmental etiology. Thyroid. 2000;10:791–798.
38. Ban, Y, Tomer, Y. Susceptibility genes in thyroid autoimmunity. Clin Dev Immunol. 2005;12:47–58.
39. Tomer, Y, Menconi, F, Davies, TF, et al. Dissecting genetic heterogeneity in autoimmune thyroid diseases by subset analysis. J Autoimmun. 2007;29:69–77.
40. Vladutiu, AO, Rose, NR. Autoimmune murine thyroiditis: relation to histocompatibility (H-2) type. Science. 1971;174:1137–1139.
41. Farid, NR, Barnard, JM, Marshall, WH. The association of HLA with autoimmune thyroid disease in Newfoundland. The influence of HLA homozygosity in Graves’ disease. Tissue Antigens. 1976;8:181–189.
42. Irvine, WJ, Gray, RS, Morris, PJ, et al. Correlation of HLA and thyroid antibodies with clinical course of thyrotoxicosis treated with antithyroid drugs. Lancet. 1977;2:898–900.
43. Tomer, Y, Davies, TF. Searching for the autoimmune thyroid disease susceptibility genes: from gene mapping to gene function. Endoc Rev. 2003;24:694–717.
44. Jacobson, EM, Huber, A, Tomer, Y. The HLA gene complex in thyroid autoimmunity: from epidemiology to etiology. J Autoimmun. 2008;30:58–62.
45. Farid, NR, Barnard, JM, Marshall, WH. The association of HLA with autoimmune thyroid disease in Newfoundland. The influence of HLA homozygosity in Graves’ disease. Tissue Antigens. 1976;8:181–189.
46. Banchereau, J, Bazan, F, Blanchard, D, et al. The CD40 antigen and its ligand. Annu Rev Immunol. 1994;12:881–922.
47. Teft, WA, Kirchhof, MG, Madrenas, J. A molecular perspective of CTLA-4 function. Annu Rev Immunol. 2006;24:65–97.
47a. Tomer, Y, Greenberg, DA, Barbesino, G, et al. CTLA-4 and not CD28 is a susceptibility gene for thyroid autoantibody production. J Clin Endocrinol Metab. 2001;86:1687–1693.
47b. Ueda, H, Howson, JM, Esposito, L, et al. Association of the T-cell regulatory gene CTLA4 with suceptibility to autoimmune disease. Nature. 2003;423:506–511.
48. Mayans, S, Lackovic, K, Nyholm, C, et al. CT60 genotype does not affect CTLA-4 isoform expression despite association to T1D and AITD in northern Sweden. BMC Med Genet. 2007;8:3.
49. Velaga, MR, Wilson, V, Jennings, CE, et al. The codon 620 tryptophan allele of the lymphoid tyrosine phosphatase (LYP) gene is a major determinant of Graves’ disease. J Clin Endocrinol Metab. 2004;89:5862–5865.
50. Criswell, LA, Pfeiffer, KA, Lum, RF, et al. Analysis of families in the multiple autoimmune disease genetics consortium (MADGC) collection: the PTPN22 620W allele associates with multiple autoimmune phenotypes. Am J Hum Genet. 2005;76:561–571.
51. Tomer, Y, Greenberg, DA, Conception, E, et al. Thyroglobulin is a thyroid specific gene for the familial autoimmune diseases. J Clin Endocrinol Metab. 2002;87:404–407.
52. Ban, Y, Greenberg, DA, Concepcion, E, et al. Amino acid substitutions in the thyroglobulin gene are associated with susceptibility to human and murine autoimmune thyroid disease. Proc Natl Acad Sci U S A. 2003;100:15119–15124.
53. Prummel, MF, Strieder, T, Wiersinga, WM. The environment and autoimmune thyroid diseases. Eur J Endocrinol. 2004;150:605–618.
54. Brix, TH, Christensen, K, Niels, VH, et al. Genetic versus environment in Graves’ disease—a population based twin study. Thyroid. 1997;7(suppl):13.
55. Tomer, Y, Davies, TF. Infection, thyroid disease and autoimmunity. Endocr Rev. 1993;14:107–120.
56. Phillips, DI, Barker, DJ, Rees, SB, et al. The geographical distribution of thyrotoxicosis in England according to the presence or absence of TSH-receptor antibodies. Clin Endocrinol (Oxf). 1985;23:283–287.
57. Cox, SP, Phillips, DIW, Osmond, C. Does infection initiate Graves disease? A population based 10 year study. Autoimmunity. 1989;4:43–49.
58. Facciani, JM, Kazim, M. Absence of seasonal variation in Graves disease. Ophthalm Plast Reconstr Surg. 2000;16:67–71.
59. Toft, AD, Blackwell, CC, Saadi, AT, et al. Secretor status and infection in patients with Grave's disease. Autoimmunity. 1990;7:279–289.
60. Prabhkar, BS, Bhan, RS, Smith, TJ. Current perspective on the pathogenesis of Graves’ disease and ophthalmopathy. Endocr Rev. 2003;24:802–835.
61. Shenkman, L, Bottone, EJ. Antibodies to Yersinia enterocolitica in thyroid disease. Ann Intern Med. 1976;85:735–739.
62. Lidman, K, Eriksson, U, Norberg, R, et al. Indirect immunofluorescence staining of human thyroid by antibodies occurring in Yersinia enterocolitica infections. Clin Exp Immunol. 1976;23:429–435.
63. Bech, K. Yersinia enterocolitica and thyroid autoimmunity. Autoimmunity. 1990;7:291–294.
64. Brix, TH, Hansen, PS, Hegedüs, L, et al. Too early to dismiss Yersinia enterocolitica infection in the aetiology of Graves’ disease. Evidence from a twin case-control study. Clin Endocrinol. 2008.
65. Weiss, M, Ingbar, SH, Winblad, S, et al. Demonstration of a saturable binding site for thyrotropin in Yersinia enterocolitica. Science. 1983;219:1331–1333.
66. Heyma, P, Harrison, LC, Robinsk-Browne, R. Thyrotrophin (TSH) binding sites on Yersinia enterocolitica recognized by immunoglobulins from humans with Graves’ disease. Clin Exp Immunol. 1986;64:249–254.
67. Wenzel, BE, Heesemann, J, Heufelder, A, et al. Enteropathogenic Yersinia enterocolitica and organ-specific autoimmune diseases in man. Contrib Microbiol Immunol. 1991;12:80–88.
68. Luo, G, Fan, JL, Seetharamaiah, GS, et al. Immunization of mice with Yersinia enterocolitica leads to the induction of antithyrotropin receptor antibodies. J Immunol. 1993;151:922–928.
69. Lindholm, H, Visakorpi, R. Late complications after a Yersinia enterocolitica epidemic: a follow up study. Ann Rheum Dis. 1991;50:694–696.
70. Kohn, LD, Napolitano, G, Singer, DS, et al. Graves’ disease: a host defense mechanism gone awry. Int Rev Immunol. 2000;19:633–664.
71. Ciampolillo, A, Mirakian, R, Schulz, T, et al. Retrovirus-like sequences in Graves’ disease: implications for human autoimmunity. Lancet. 1989;1:1096–1099.
72. Humphrey, M, Baker, JJ, Carr, FE, et al. Absence of retroviral sequences in Graves’ disease. Lancet. 1991;337:17–18.
73. Fierabracci, A, Upton, CP, Hajibagheri, N, et al. Lack of detection of retroviral particles (HIAP-1) in the H9 T cell line co-cultured with thyrocytes of Graves’ disease. J Autoimmun. 2001;16:457–462.
74. Wick, G, Grubeck-Loebenstein, B, Trieb, K, et al. Human foamy virus antigens in thyroid tissue of Graves’ disease patients. Int Arch Allergy Immunol. 1992;99:153–156.
75. Schweizer, M, Turek, R, Reinhardt, M, et al. Absence of foamy virus DNA in Graves’ disease. AIDS Res Hum Retroviruses. 1994;10:601–605.
76. Schweizer, M, Turek, R, Hahn, H, et al. Markers of foamy virus infections in monkeys, apes, and accidentally infected humans: Appropriate testing fails to confirm suspected foamy virus prevalence in humans. AIDS Res Hum Retroviruses. 1995;11:161–170.
77. Burch, HB, Nagy, EV, Lukes, YG, et al. Nucleotide and amino acid homology between the human thyrotropin receptor and HIV-1 nef protein: identification and functional analysis. Biochem Biophys Res Commun. 1991;181:498–505.
78. Tas, M, de Haan-Meulman, M, Kabel, PJ, et al. Defects in monocyte polarization and dendritic cell clustering in patients with Graves’ disease. A putative role for a non-specific immunoregulatory factor related to retroviral p15E. Clin Endocrinol. 1991;34:441–448.
79. Leib-Mosch, C, Bachmann, M, Brack-Werner, R, et al. Expression and biological significance of human endogenous retroviral sequences. Leukemia. 1992;6(suppl):72–75.
80. Jaspan, JB, Luo, H, Ahmed, B, et al. Evidence for a retroviral trigger in Graves’ disease. Autoimmunity. 1995;20:135–142.
81. Fierabracci, A, Upton, CP, Hajibagheri, N, et al. Lack of detection of retroviral particles (HIAP-1) in the H9 T cell line co-cultured with thyrocytes of Graves’ disease. J Autoimmunity. 2001;16:457–462.
82. Fierabracci, A, Hammond, L, Lowdell, M, et al. The effect of staphylococcal enterotoxin B on thyrocyte HLA molecule expression. J Autoimmun. 1999;12:305–314.
83. Kondrashova, A, Viskari, H, Haapala, A-M, et al. Serological evidence of thyroid autoimmunity among schoolchildren in two different socioeconomic environments. J Clin Endocrinol Metab. 2008;93:729–734.
84. Chiovato, L, Pinchera, A. Stressful life events and Graves’ disease. Eur J Endocrinol. 1996;134:680–682.
85. Bagnasco, M, Bossert, I, Pesce, G. Stress and autoimmune thyroid diseases. Neuroimmunomodulation. 2007;13:309–317.
86. Mizokami, T, Wu Li, A, El-Kaissi, S, et al. Stress and thyroid autoimmunity. Thyroid. 2004;14:1047–1055.
87. Dayan, CM. Stressful life events and Graves’ disease revisited. Clin Endocrinol. 2001;55:15–19.
88. Chiovato, L, Marino, M, Perugi, G, et al. Chronic recurrent stress due to panic disorder does not precipitate Graves’ disease. J Endocrinol Invest. 1998;21:758–764.
89. Stein, SP, Keller, SE, Schleifer, SJ. Stress and immunomodulation: the role of depression and neuroendocrine function. J Immunol. 1985;135(suppl):827–833.
90. Ziemssen, T, Kern, S. Psychoneuroimmunology—cross-talk between the immune and nervous systems. J Neurol Suppl. 2007;2:II8–11.
91. Amino, N, Tada, H, Hidaka, Y. Postpartum autoimmune thyroid syndrome: a model of aggravation of autoimmune disease. Thyroid. 1999;9:705–713.
92. Wong, GW, Kwok, MY, Ou, Y. High incidence of juvenile Graves’ disease in Hong Kong. Clin Endocrinol (Oxf). 1995;43:697–700.
93. Zandman-Goddard, G, Peeva, E, Shoenfeld, Y. Gender and autoimmunity. Autoimmun Rev. 2007;2007(6):366–372.
94. Chiovato, L, Lapi, P, Fiore, E, et al. Thyroid autoimmunity and female gender. J Endocrinol Invest. 1993;16:384–391.
95. Lutfi, RJ, Fridmanis, M, Misiunas, AL, et al. Association of melasma with thyroid autoimmunity and other thyroidal abnormalities and their relationship to the origin of the melasma. J Clin Endocrinol Metab. 1985;61:28–31.
96. Chiovato, L, Larizza, D, Bendinelli, G, et al. Autoimmune hypothyroidism and hyperthyroidism in patients with Turner's syndrome. Eur J Endocrinol. 1996;134:568–575.
97. Vallotton, MB, Forbes, AP. Autoimmunity in gonadal dysgenesis and Klinefelter's syndrome. Lancet. 1967;1:648–651.
98. Rotondi, M, Pirali, B, Lodigiani, S, et al. The postpartum period and the onset of Graves’ disease: an overestimated risk factor. Eur J Endocrinol. 2008;159:161–165.
99. Rotondi, M, Cappelli, C, Pirali, B, et al. The effect of pregnancy on subsequent relapse from Graves’ disease following a successful course of anti-thyroid drug therapy. J Clin Endocrinol Metab. 2008.
100. Davies, TF. The thyroid immunology of the post-partum period. Thyroid. 1999;9:675–684.
101. Yin, X, Latif, R, Tomer, Y, et al. Thyroid epigenetics: X chromosome inactivation in patients with autoimmune thyroid disease. Ann N Y Acad Sci. 2007;1110:193–200.
102. Bartalena, L. Smoking and Graves’ disease. J Endocrinol Invest. 2002;25:402.
103. Holm, IA, Manson, JE, Michels, KB, et al. Smoking and other lifestyle factors and the risk of Graves’ hyperthyroidism. Arch Intern Med. 2005;165:1606–1611.
104. Monzani, F, Del Guerra, P, Caraccio, N, et al. Appearance of Graves’ disease after percutaneous ethanol injection for the treatment of hyperfunctioning thyroid adenoma. J Endocrinol Invest. 1977;20:294–298.
105. Chiovato, L, Santini, F, Vitti, P, et al. Appearance of thyroid stimulating antibody and Graves’ disease after radioiodine therapy for toxic nodular goitre. Clin Endocrinol (Oxf). 1994;40:803–806.
106. LiVolsi, VA. The pathology of autoimmune thyroid disease: a review. Thyroid. 1994;4:333–339.
107. Davies, TF, Ando, T, Lin, RY, et al. Thyrotropin receptor-associated diseases: from adenomata to Graves disease. J Clin Invest. 2005;115:1972–1983.
108. Rapoport, B, McLachlan, SM. The thyrotropin receptor in Graves’ disease. Thyroid. 2007;17:911–922.
109. Rapoport, B, Chazenbalk, GD, Jaume, JC, et al. The thyrotropin (TSH) receptor: interaction with TSH and autoantibodies. Endocr Rev. 1998;19:673–716.
110. Rees Smith, B, McLachlan, SM, Furmaniak, J. Autoantibodies to the thyrotropin receptor. Endocr Rev. 1988;9:106–121.
111. McKenzie, JM, Zakarija, M. Clinical review 3: the clinical use of thyrotropin receptor antibody measurements. J Clin Endocrinol Metab. 1989;69:1093–1096.
112. Chiovato, L, Vitti, P, Bendinelli, G, et al. Detection of antibodies blocking thyrotropin effect using Chinese hamster ovary cells transfected with the cloned human TSH receptor. J Endocrinol Invest. 1994;17:809–816.
113. Chazenbalk, GD, Pichurin, P, Chen, CR, et al. Thyroid-stimulating autoantibodies in Graves disease preferentially recognize the free A subunit, not the thyrotropin holoreceptor. J Clin Invest. 2002;110:209–217.
114. Chen, CR, Pichurin, P, Nagayama, Y, et al. The thyrotropin receptor autoantigen in Graves disease is the culprit as well as the victim. J Clin Invest. 2003;111:1897–1904.
115. Chen, CR, Tanaka, K, Chazenbalk, GD, et al. A full biological response to autoantibodies in Graves’ disease requires a disulfide-bonded loop in the thyrotropin receptor N terminus homologous to a laminin epidermal growth factor-like domain. J Biol Chem. 2001;276:14767–14772.
116. Cundiff, JG, Kaithamana, S, Seetharamaiah, GS, et al. Studies using recombinant fragments of human TSH receptor reveal apparent diversity in the binding specificities of antibodies that block TSH binding to its receptor or stimulate thyroid hormone production. J Clin Endocrinol Metab. 2001;86:4254–4260.
117. Tahara, K, Ishikawa, N, Yamamoto, K, et al. Epitopes for thyroid stimulating and blocking autoantibodies on the extracellular domain of the human thyrotropin receptor. Thyroid. 1997;7:867–877.
118. Schwarz-Lauer, L, Chazenbalk, GD, Mclachlan, SM, et al. Evidence for a simplified view of autoantibody interactions with the thyrotropin receptor. Thyroid. 2002;12:115–120.
119. McKenzie, JM. Delayed thyroid response to serum from thyrotoxic patients. Endocrinology. 1958;62:865–868.
120. Schott, M, Scherbaum, WA, Morgenthaler, NG. Thyrotropin receptor autoantibodies in Graves’ disease. Trends Endocrinol Metab. 2005;16:243–248.
121. Shewring, G, Smith, BR. An improved radioreceptor assay for TSH receptor antibodies. Clin Endocrinol (Oxf). 1982;17:409–417.
122. Southgate, K, Creagh, F, Teece, M, et al. A receptor assay for the measurement of TSH receptor antibodies in unextracted serum. Clin Endocrinol (Oxf). 1984;20:539–548.
123. Filetti, S, Foti, D, Costante, G, et al. Recombinant human TSH receptor in a radioreceptor assay for the measurement of TSH receptor autoantibodies. J Clin Endocrinol Metab. 1991;72:1096–1101.
124. Costagliola, S, Swillens, S, Niccoli, P, et al. Binding assay for thyrotropin receptor autoantibodies using the recombinant receptor protein. J Clin Endocrinol Metab. 1992;75:1540–1544.
125. Ludgate, M, Costagliola, S, Danguy, D, et al. Recombinant TSH-receptor for determination of TSH-receptor-antibodies. Exp Clin Endocrinol. 1992;100:73–74.
126. Costagliola, S, Morgenthaler, NG, Hoermann, R, et al. Second generation assay for thyrotropin receptor antibodies has superior diagnostic sensitivity for Graves’ disease. J Clin Endocrinol Metab. 1999;84:90–97.
127. Bolton, J, Sanders, J, Oda, Y, et al. Measurement of thyroid-stimulating hormone receptor autoantibodies by ELISA. Clin Chem. 1999;45:2285–2287.
128. Rapoport, B, Greenspan, FS, Filetti, S, et al. Clinical experience with a human thyroid cell bioassay for thyroid-stimulating immunoglobulin. J Clin Endocrinol Metab. 1984;58:332–338.
129. Vitti, P, Valente, WA, Ambesi-Impiombato, FS, et al. Graves’ IgG stimulation of continuously cultured rat thyroid cells: a sensitive and potentially useful clinical assay. J Endocrinol Invest. 1982;5:179–182.
130. Vitti, P, Rotella, CM, Valente, WA, et al. Characterization of the optimal stimulatory effects of Graves’ monoclonal and serum immunoglobulin G on adenosine 3′,5′-monophosphate production in fRTL-5 thyroid cells: a potential clinical assay. J Clin Endocrinol Metab. 1983;57:782–791.
131. Ludgate, M, Perret, J, Parmentier, M, et al. Use of the recombinant human thyrotropin receptor (TSH-R) expressed in mammalian cell lines to assay TSH-R autoantibodies. Mol Cell Endocrinol. 1990;73:R13–R18.
132. Vitti, P, Elisei, R, Tonacchera, M, et al. Detection of thyroid-stimulating antibody using Chinese hamster ovary cells transfected with cloned human thyrotropin receptor. J Clin Endocrinol Metab. 1993;76:499–503.
133. Morgenthaler, NG, Pampel, I, Aust, G, et al. Application of a bioassay with CHO cells for the routine detection of stimulating and blocking autoantibodies to the TSH-receptor. Horm Metab Res. 1998;30:162–168.
134. Watson, PF, Ajjan, RA, Phipps, J, et al. A new chemiluminescent assay for the rapid detection of thyroid stimulating antibodies in Graves’ disease. Clin Endocrinol (Oxf). 1998;49:577–581.
135. Evans, C, Morgenthaler, NG, Lee, S, et al. Development of a luminescent bioassay for thyroid stimulating antibodies. J Clin Endocrinol Metab. 1999;84:374–377.
136. Minich, WB, Lenzner, C, Bergmann, A, et al. A coated tube assay for the detection of blocking thyrotropin receptor autoantibodies. J Clin Endocrinol Metab. 2004;89:352–356.
137. McKenzie, JM. The gamma globulin of Graves’ disease: thyroid stimulation by fraction and fragment. Trans Assoc Am Physicians. 1965;78:174–186.
138. Kriss, JP, Pleshakov, V, Rosenblum, AL, et al. Studies on the pathogenesis of the ophthalmopathy of Graves’ disease. J Clin Endocrinol Metab. 1967;27:582–593.
139. Pinchera, A, Liberti, P, De Santis, R, et al. Relationship between the long-acting thyroid stimulator and circulating thyroid antibodies in Graves’ disease. J Clin Endocrinol Metab. 1967;27:1758–1760.
140. McKenzie, JM, Zakarija, M. Fetal and neonatal hyperthyroidism and hypothyroidism due to maternal TSH receptor antibodies. Thyroid. 1992;2:155–159.
141. Leovey, A, Nagy, E, Balazs, G, et al. Lymphocytes resided in the thyroid are the main source of TSH-receptor antibodies in Basedow's-Graves’ disease? Exp Clin Endocrinol. 1992;99:147–150.
142. McLachlan, SM, Dickinson, AM, Malcolm, A, et al. Thyroid autoantibody synthesis by cultures of thyroid and peripheral blood lymphocytes. I: Lymphocyte markers and response to pokeweed mitogen. Isr J Med Sci. 1983;52:45–53.
143. Okuda, J, Akamizu, T, Sugawa, H, et al. Preparation and characterization of monoclonal antithyrotropin receptor antibodies obtained from peripheral lymphocytes of hypothyroid patients with primary myxedema. J Clin Endocrinol Metab. 1994;79:1600–1604.
144. Morgenthaler, NG, Kim, MR, Tremble, J, et al. Human immunoglobulin G autoantibodies to the thyrotropin receptor from Epstein-Barr virus–transformed B lymphocytes: characterization by immunoprecipitation with recombinant antigen and biological activity. J Clin Endocrinol Metab. 1996;81:3155–3161.
145. Sugenoya, A, Kobayashi, S, Kasuga, Y, et al. Evidence of intrathyroidal accumulation of TSH receptor antibody in Graves’ disease. Acta Endocrinol (Copenh). 1992;126:416–418.
146. Pinchera, A, Liberti, P, Martino, E, et al. Effects of antithyroid therapy on the long-acting thyroid stimulator and the antithyroglobulin antibodies. J Clin Endocrinol. 1969;29:231–289.
147. Fenzi, GF, Hashizume, K, Roudeboush, CP, et al. Changes in thyroid-stimulating immunoglobulins during antithyroid therapy. J Clin Endocrinol Metab. 1979;48:572–576.
148. Weetman, AP, McGregor, AM, Hall, R. Evidence for an effect of antithyroid drugs on the natural history of Graves’ disease. Clin Endocrinol. 1984;21:163–172.
149. Tsui, S, Naik, V, Hoa, N, et al. Evidence for an association between thyroid-stimulating hormone and insulin-like growth factor 1 receptors: a tale of two antigens implicated in Graves’ disease. J Immunol. 2008;181:4397–4405.
150. Romagnani, S. Regulation of the T cell response. Clin Exp Allergy. 2006;11:1357–1366.
151. Abbas, AK, Murphy, KM, Sher, A. Functional diversity of T lymphocytes. Nature. 1996;383:787–793.
152. Dardalhon, V, Korn, T, Kuchroo, VK, et al. Role of Th1 and Th17 cells in organ-specific autoimmunity. J Autoimmun. 2008.
153. Jackson, RA, Haynes, BF, Burch, WM, et al. Ia+ T cells in new onset Graves’ disease. J Clin Endocrinol Metab. 1984;59:187–190.
154. Matsunaga, M, Eguchi, K, Fukuda, T, et al. Class II major histocompatibility complex antigen expression and cellular interactions in thyroid glands of Graves’ disease. J Clin Endocrinol Metab. 1986;62:723–728.
155. Zeki, K, Fujihira, T, Shirakawa, F, et al. Existence and immunological significance of circulating Ia+ T cells in autoimmune thyroid diseases. Acta Endocrinol (Copenh). 1987;115:282–288.
156. McLachlan, SM, Pegg, CA, Atherton, MC, et al. Subpopulations of thyroid autoantibody secreting lymphocytes in Graves’ and Hashimoto thyroid glands. Clin Exp Immunol. 1986;65:319–328.
157. Aozasa, M, Amino, N, Iwatani, Y, et al. Separation and analysis of mononuclear cells infiltrating the thyroid of patients with Graves’ disease. Clin Immunol Immunopathol. 1987;43:343–353.
158. Mariotti, S, del Prete, GF, Mastromauro, C, et al. The autoimmune infiltrate of Basedow's disease: analysis of clonal level and comparison with Hashimoto's thyroiditis. Exp Clin Endocrinol. 1991;97:139–146.
159. Martin, A, Davies, TF. T cells and human autoimmune thyroid disease: emerging data show lack of need to invoke suppressor T-cell problems. Thyroid. 1992;2:247–261.
160. Ueki, Y, Eguchi, K, Otsubo, T, et al. Phenotypic analyses and concanavalin-A–induced suppressor cell dysfunction of intrathyroidal lymphocytes from patients with Graves’ disease. J Clin Endocrinol Metab. 1988;67:1018–1024.
161. Martin, A, Goldsmith, NK, Friedman, EW, et al. Intrathyroidal accumulation of T cell phenotypes in autoimmune thyroid disease. Autoimmunity. 1990;6:269–281.
162. Londei, M, Bottazzo, GF, Feldmann, M. Human T-cell clones from autoimmune thyroid glands: specific recognition of autologous thyroid cells. Science. 1985;228:85–89.
163. Mackenzie, WA, Davies, TF. An intrathyroidal T-cell clone specifically cytotoxic for human thyroid cells. Immunology. 1987;61:101–103.
164. Mackenzie, WA, Schwartz, AE, Friedman, EW, et al. Intrathyroidal T cell clones from patients with autoimmune thyroid disease. J Clin Endocrinol Metab. 1987;64:818–824.
165. Fisfalen, ME, DeGroot, LJ, Quintans, J, et al. Microsomal antigen–reactive lymphocyte lines and clones derived from thyroid tissue of patients with Graves’ disease. J Clin Endocrinol Metab. 1988;66:776–784.
166. Dayan, CM, Londei, M, Corcoran, AE, et al. Autoantigen recognition by thyroid-infiltrating T cells in Graves’ disease. Proc Natl Acad Sci U S A. 1991;88:7415–7419.
167. Watson, PF, Pickerill, AP, Davies, R, et al. Analysis of cytokine gene expression in Graves’ disease and multinodular goiter. J Clin Endocrinol Metab. 1994;79:355–360.
168. Fisfalen, ME, Palmer, EM, Van Seventer, GA, et al. Thyrotropin-receptor and thyroid peroxidase–specific T cell clones and their cytokine profile in autoimmune thyroid disease. J Clin Endocrinol Metab. 1997;82:3655–3663.
169. Davis, MM, Bjorkman, PJ. T-cell antigen receptor genes and T-cell recognition. Nature. 1988;334:395–402.
170. Weiss, A. Structure and function of the T cell antigen receptor. J Clin Invest. 1990;86:1015–1022.
171. Davies, TF, Concepcion, ES, Ben-Nun, A, et al. T-cell receptor V gene use in autoimmune thyroid disease: direct assessment by thyroid aspiration. J Clin Endocrinol Metab. 1993;76:660–666.
172. Davies, TF, Martin, A, Concepcion, ES, et al. Evidence of limited variability of antigen receptors on intrathyroidal T cells in autoimmune thyroid disease. N Engl J Med. 1991;325:238–244.
173. Tandon, N, Freeman, MA, Weetman, AP. T cell response to synthetic TSH receptor peptides in Graves’ disease. Clin Exp Immunol. 1992;89:468–473.
174. Heufelder, AE, Wenzel, BE, Scriba, PC. Antigen receptor variable region repertoires expressed by T cells infiltrating thyroid, retroorbital, and pretibial tissue in Graves’ disease. J Clin Endocrinol Metab. 1996;81:3733–3739.
175. Nakashima, M, Kong, YM, Davies, TF. The role of T cells expressing TcR V beta 13 in autoimmune thyroiditis induced by transfer of mouse thyroglobulin-activated lymphocytes: Identification of two common CDR3 motifs. Clin Immunol Immunopathol. 1996;80:204–210.
176. McIntosh, RS, Watson, PF, Pickerill, AP, et al. No restriction of intrathyroidal T cell receptor V alpha families in the thyroid of Graves’ disease. Clin Exp Immunol. 1993;91:147–152.
177. Caso-Pelaez, E, McGregor, AM, Banga, JP. A polyclonal T cell repertoire of V-alpha and V-beta T cell receptor gene families in intrathyroidal T lymphocytes of Graves’ disease patients. Scand J Immunol. 1995;41:141–147.
178. Martin, A, Nakashima, M, Zhou, A, et al. Detection of major T cell epitopes on human thyroid stimulating hormone receptor by overriding immune heterogeneity in patients with Graves’ disease. J Clin Endocrinol Metab. 1997;82:3361–3366.
179. Inaba, H, Martin, W, De Groot, AS, et al. Thyrotropin receptor epitopes and their relation to histocompatibility leukocyte antigen-DR molecules in Graves’ disease. J Clin Endocrinol Metab. 2006;91:2286–2294.
180. Rotondi, M, Chiovato, L, Romagnani, S, et al. Role of chemokines in endocrine autoimmune diseases. Endocr Rev. 2007;28:492–520.
181. Bottazzo, GF, Todd, I, Pujol, BR. Hypotheses on genetic contributions to the etiology of diabetes mellitus. Immunol Today. 1984;5:230.
182. Davies, TF. Co-culture of human thyroid monolayer cells and autologous T cells: impact of HLA class II antigen expression. J Clin Endocrinol Metab. 1985;61:418–422.
183. Weetman, AP. The potential immunological role of the thyroid cell in autoimmune thyroid disease. Thyroid. 1994;4:493–499.
184. Hanafusa, T, Pujol, BR, Chiovato, L, et al. Aberrant expression of HLA-DR antigen on thyrocytes in Graves’ disease: relevance for autoimmunity. Lancet. 1983;2:1111–1115.
185. Bottazzo, GF, Pujol, BR, Hanafusa, T, et al. Role of aberrant HLA-DR expression and antigen presentation in induction of endocrine autoimmunity. Lancet. 1983;2:1115–1119.
186. Jansson, R, Karlsson, A, Forsum, U. Intrathyroidal HLA-DR expression and T lymphocyte phenotypes in Graves’ thyrotoxicosis, Hashimoto's thyroiditis and nodular colloid goiter. Clin Exp Immunol. 1984;58:264–272.
187. Aichinger, G, Fill, H, Wick, G. In situ immune complexes, lymphocyte subpopulations, and HLA-DR–positive epithelial cells in Hashimoto thyroiditis. Lab Invest. 1985;52:132–140.
188. Kawakami, Y, Kuzuya, N, Watanabe, T, et al. Induction of experimental thyroiditis in mice by recombinant interferon gamma administration. Acta Endocrinol (Copenh). 1990;122:41–48.
189. Tomer, Y, Blackard, JT, Akeno, N. Interferon alpha treatment and thyroid dysfunction. Endocrinol Metab Clin North Am. 2007;36:105110–105166.
190. Caturegli, P, Hejazi, M, Suzuki, K, et al. Hypothyroidism in transgenic mice expressing IFN-gamma in the thyroid. Proc Natl Acad Sci U S A. 2000;97:1719–1724.
191. Alimi, E, Huang, S, Brazillet, MP, et al. Experimental autoimmune thyroiditis (EAT) in mice lacking the IFN-gamma receptor gene. Eur J Immunol. 1998;28:201–208.
192. Voorby, HAM, Kabel, PJ, De Haan, M, et al. Dendritic cells and class II MHC expression on thyrocytes during the autoimmune thyroid disease of the BB rat. Clin Immunol Immunopathol. 1990;55:9–22.
193. Davies, TF. The complex role of epithelial cell MHC class II antigen expression in autoimmune thyroid disease. Autoimmunity. 1990;8:87–89.
194. Londei, M, Lamb, JR, Bottazzo, GF, et al. Epithelial cells expressing aberrant MHC class II determinants can present antigen to cloned human T cells. Nature. 1984;312:639–641.
195. Kimura, H, Davies, TF. Thyroid-specific T cells in the normal Wistar rat. II: T cell clones interact with cloned Wistar rat thyroid cells and provide direct evidence for autoantigen presentation by thyroid epithelial cells. Clin Immunol Immunopathol. 1991;58:195–206.
196. Eguchi, K, Otsubo, T, Kawabe, K, et al. The remarkable proliferation of helper T cell subset in response to autologous thyrocytes and intrathyroidal T cells from patients with Graves’ disease. Isr J Med Sci. 1987;70:403–410.
197. Kong, YC, Flynn, JC, Wan, Q, et al. HLA and H2 class II transgenic mouse models to study susceptibility and protection in autoimmune thyroid disease. Autoimmunity. 2003;36:397–404.
198. Muixí, L, Carrascal, M, Alvarez, I, et al. Thyroglobulin peptides associate in vivo to HLA-DR in autoimmune thyroid glands. J Immunol. 2008;181:795–807.
199. Romagnani, P, Rotondi, M, Lazzeri, E, et al. Expression of IP-10/CXCL10 and MIG/CXCL9 in the thyroid and increased levels of IP-10/CXCL10 in the serum of patients with recent onset Graves’ disease. Am J Pathol. 2002;161:195–206.
200. Hutchings, P, Rayner, DC, Champion, BR, et al. High efficiency antigen presentation by thyroglobulin-primed murine spleen B cells. Eur J Immunol. 1987;17:393–398.
201. Kabel, PJ, Voorbij, HA, De Haan, M, et al. Intrathyroidal dendritic cells. J Clin Endocrinol Metab. 1988;66:199–207.
202. Salmaso, C, Olive, D, Pesce, G, et al. Costimulatory molecules and autoimmune thyroid diseases. Autoimmunity. 2002;35:159–167.
203. Battifora, M, Pesce, G, Paolieri, F, et al. B7.1 costimulatory molecule is expressed on thyroid follicular cells in Hashimoto's thyroiditis, but not in Graves’ disease. J Clin Endocrinol Metab. 1998;83:4130–4139.
204. Weetman, AP, McGregor, AM. Autoimmune thyroid disease: further developments in our understanding. Endocr Rev. 1994;15:788–830.
205. Shimojo, N, Kohno, Y, Yagamuchi, K, et al. Induction of Graves-like disease in mice by immunization with fibroblasts transfected with the thyrotropin receptor and a class II molecule. Proc Natl Acad Sci U S A. 1996;93:11074–11079.
206. McLachlan, SM, Nagayama, Y, Rapoport, B. Insight into Graves’ hyperthyroidism from animal models. Endocr Rev. 2005;26:800–832.
207. Nagayama, Y. Graves’ animal models of Graves’ hyperthyroidism. Thyroid. 2007;17:981–988.
208. Brent, GA. Graves’ disease. N Engl J Med. 2008;358:2594–2605.
209. Cooper, DS. Hyperthyroidism. Lancet. 2003;362:459–468.
210. Leznoff, A, Sussman, GL. Syndrome of idiopathic chronic urticaria and angioedema with thyroid autoimmunity: a study of 90 patients. J Allergy Clin Immunol. 1989;84:66–71.
211. Betterle, C, Dal Pra, C, Mantero, F, et al. Autoimmune adrenal insufficiency and autoimmune polyendocrine syndromes: autoantibodies, autoantigens, and their applicability in diagnosis and disease prediction. Endocr Rev. 2002;23:327–364.
212. Kahaly, GJ, Dillmann, WH. Thyroid hormone action in the heart. Endocr Rev. 2005;26:704–728.
213. Klein, I, Danzi, S. Thyroid disease and the heart. Circulation. 2007;116:1725–1735.
214. Klein, I, Ojamaa, K. Thyrotoxicosis and the heart. Endocrinol Metab Clin North Am. 1998;27:51–62.
215. Shafer, RB, Prentiss, RA, Bond, JH. Gastrointestinal transit in thyroid disease. Gastroenterology. 1984;86:852–855.
216. Thomas, FB, Caldwell, JH, Greenberger, NJ. Steatorrhea in thyrotoxicosis. Relation to hypermotility and excessive dietary fat. Ann Intern Med. 1973;78:669–675.
217. Kubota, S, Amino, N, Matsumoto, Y, et al. Serial changes in liver function tests in patients with thyrotoxicosis induced by Graves’ disease and painless thyroiditis. Thyroid. 2008;18:283–287.
218. Bunevicius, R, Prange, AJ, Jr. Psychiatric manifestations of Graves’ hyperthyroidism: pathophysiology and treatment options. CNS Drugs. 2006;20:897–909.
219. Mariotti, S, Franceschi, C, Cossarizza, A, et al. The aging thyroid. Endocr Rev. 1995;16:686–715.
220. Feibel, JH, Campa, JF. Thyrotoxic neuropathy (Basedow's paraplegia). J Neurol Neurosurg Psychiatry. 1976;39:491–497.
221. Pandit, L, Shankar, SK, Gayathri, N, et al. Acute thyrotoxic neuropathy—Basedow's paraplegia revisited. J Neurol Sci. 1998;155:211–214.
222. Marino, M, Ricciardi, R, Pinchera, A, et al. Mild clinical expression of myasthenia gravis associated with autoimmune thyroid diseases. J Clin Endocrinol Metab. 1997;82:438–443.
223. Marino, M, Barbesino, G, Manetti, L, et al. Mild clinical expression of myasthenia gravis associated with autoimmune thyroid disease (letter). J Clin Endocrinol Metab. 1997;82:3905–3906.
224. Marinò, M, Barbesino, G, Pinchera, A, et al. Increased frequency of euthyroid ophthalmopathy in patients with Graves’ disease associated with myasthenia gravis. Thyroid. 2000;10:799–802.
225. Kanazawa, M, Shimohata, T, Tanaka, K, et al. Clinical features of patients with myasthenia gravis associated with autoimmune diseases. Eur J Neurol. 2007;14:1403–1404.
226. Kung, AW. Clinical review: thyrotoxic periodic paralysis: a diagnostic challenge. J Clin Endocrinol Metab. 2006;91:2490–2495.
227. Tamai, H, Tanaka, K, Komaki, G, et al. HLA and thyrotoxic periodic paralysis in Japanese patients. J Clin Endocrinol Metab. 1987;64:1075–1078.
228. Dias Da Silva, MR, Cerutti, JM, Arnaldi, LA, et al. A mutation in the KCNE3 potassium channel gene is associated with susceptibility to thyrotoxic hypokalemic periodic paralysis. J Clin Ebndocrinol Metab. 2002;87:4879–4880.
229. Painter, SE, Kleerekoper, M, Camacho, PM. Secondary osteoporosis: a review of the recent evidence. Endocr Pract. 2006;12:436–445.
230. Vestergaard, P, Mosekilde, L. Hyperthyroidism, bone mineral, and fracture risk—a meta-analysis. Thyroid. 2003:585–593.
231. Wexler, JA, Sharretts, J. Thyroid and bone. Endocrinol Metab Clin North Am. 2007;36:673–705.
232. Franklyn, JA, Maisonneuve, P, Sheppard, MC, et al. Mortality after the treatment of hyperthyroidism with radioactive iodine. N Engl J Med. 1998;338:712–718.
233. Abe, E, Marians, RC, Yu, W, et al. TSH is a negative regulator of skeletal remodeling. Cell. 2003;115:151–162.
234. Sun, L, Vukicevic, S, Baliram, R, et al. Intermittent recombinant TSH injections prevent ovariectomy-induced bone loss. Proc Natl Acad Sci U S A. 2008;105:4289–4294.
235. Zaidi, M, Sun, L, Davies, TF, et al. Low TSH triggers bone loss: fact or fiction? Thyroid. 2006;16:1075–1076.
236. Hase, H, Ando, T, Eldeiry, L, et al. TNF alpha mediates the skeletal effects of thyroid-stimulating hormone. Proc Natl Acad Sci U S A. 2006;103:12849–12854.
237. Cooper, DS. Antithyroid drugs. N Engl J Med. 2005;352:905–917.
238. Fein, HG, Rivlin, RS. Anemia in thyroid diseases. Med Clin North Am. 1975;59:1133–1145.
239. Sibilla, R, Santaguida, MG, Virili, C, et al. Chronic unexplained anaemia in isolated autoimmune thyroid disease or associated with autoimmune related disorders. Clin Endocrinol (Oxf). 2008;68:640–645.
240. Aydin, Y, Berker, D, Ustün, I, et al. A very rare cause of aplastic anemia: Graves disease. South Med J. 2008;101:666–667.
241. Erem, C, Ersoz, HO, Karti, SS, et al. Blood coagulation and fibrinolysis in patients with hyperthyroidism. J Endocrinol Invest. 2002;25:345–350.
242. Krassas, GE. Thyroid disease and female reproduction. Fertil Steril. 2000;74:1063–1070.
243. Koutras, DA. Disturbances of menstruation in thyroid disease. Ann N Y Acad Sci. 1997;816:280–284.
244. Chiovato, L, Lapi, P, Fiore, E, et al. Thyroid autoimmunity and female gender. J Endocrinol Invest. 1993;16:384–391.
245. Pugeat, M, Crave, JC, Tourniaire, J, et al. Clinical utility of sex hormone-binding globulin measurement. Horm Res. 1996;45:148–155.
246. Meikle, AW. The interrelationships between thyroid dysfunction and hypogonadism in men and boys. Thyroid. 2004;14(Suppl 1):S17–25.
247. Kim, B. Thyroid hormone as a determinant of energy expenditure and the basal metabolic rate. Thyroid. 2008;18:141–144.
248. Simonides, WS, van Hardeveld, C. Thyroid hormone as a determinant of metabolic and contractile phenotype of skeletal muscle. Thyroid. 2008;18:205–216.
249. Chidakel, A, Mentuccia, D, Celi, FS. Peripheral metabolism of thyroid hormone and glucose homeostasis. Thyroid. 2005;15:899–903.
250. Duntas, LH. Thyroid disease and lipids. Thyroid. 2002;12:287–293.
251. Cachefo, A, Boucher, P, Vidon, C, et al. Hepatic lipogenesis and cholesterol synthesis in hyperthyroid patients. J Clin Endocrinol Metab. 2001;86:5353–5357.
252. Iglesias, P, Díez, JJ. Influence of thyroid dysfunction on serum concentrations of adipocytokines. Cytokine. 2007;40:61–70.
253. Feldt-Rasmussen, U. Thyroid and leptin. Thyroid. 2007;17:413–419.
254. Santini, F, Marsili, A, Mammoli, C, et al. Serum concentrations of adiponectin and leptin in patients with thyroid dysfunctions. J Endocrinol Invest. 2004;27:RC5–7.
255. Bartalena, L, Pinchera, A, Marcocci, C. Management of Graves’ ophthalmopathy: reality and perspectives. Endocr Rev. 2000;21:168–199.
256. Bartalena, L, Wiersinga, WM, Pinchera, A. Graves’ ophthalmopathy: state of the art and perspectives. J Endocrinol Invest. 2004;27:295–301.
257. Marcocci, C, Pinchera, A, Marinò, M. A treatment strategy for Graves’ orbitopathy. Nat Clin Pract Endocrinol Metab. 2007;3:430–436.
258. Fatourechi, V. Pretibial myxedema: pathophysiology and treatment options. Am J Clin Dermatol. 2005;6:295–309.
259. Rapoport, B, Alsabeh, R, Afterggod, D, et al. Elephantiasic pretibial myxedema: insight into and a hypothesis regarding the pathogenesis of the extrathyroidal manifestations of Graves’ disease. Thryoid. 2000;10:629–630.
260. Heufelder, AE, Bahn, RS, Scriba, PC. Analysis of T-cell antigen receptor variable region gene usage in patients with thyroid-related pretibial dermopathy. J Invest Dermatol. 1995;105:372–378.
261. Chang, TC, Wu, SL, Hsiao, YL, et al. TSH and TSH receptor antibody–binding sites in fibroblasts of pretibial myxedema are related to the extracellular domain of entire TSH receptor. Clin Immunol Immunopathol. 1994;71:113–120.
262. Heufelder, AE, Dutton, CM, Sarkar, G, et al. Detection of TSH receptor RNA in cultured fibroblasts from patients with Graves’ ophthalmopathy and pretibial dermopathy. Thyroid. 1993;3:297–300.
263. Stadlmayr, W, Spitzweg, C, Bichlmair, AM, et al. TSH receptor transcripts and TSH receptor–like immunoreactivity in orbital and pretibial fibroblasts of patients with Graves’ ophthalmopathy and pretibial myxedema. Thyroid. 1997;7:3–12.
264. Daumerie, C, Ludgate, M, Costagliola, S, et al. Evidence for thyrotropin receptor immunoreactivity in pretibial connective tissue from patients with thyroid-associated dermopathy. Eur J Endocrinol. 2002;146:35–38.
265. Marinò, M, Chiovato, L, Lisi, S, et al. Role of thyroglobulin in the pathogenesis of Graves’ ophthalmopathy: the hypothesis of Kriss revisited. J Endocrinol Invest. 2004;27:230–236.
266. Fatourechi, V, Bartley, GB, Eghbali-Fatourechi, GZ, et al. Graves’ dermopathy and acropachy are markers of severe Graves’ ophthalmopathy. Thyroid. 2003;13:1141–1144.
267. Dufour, DR. Laboratory tests of thyroid function: uses and limitations. Endocrinol Metab Clin North Am. 2007;36:579–594.
268. Grüters, A. Thyroid hormone transporter defects. Endocr Dev. 2007;10:118–126.
269. Mariotti, S, Martino, E, Cupini, C, et al. Low serum thyroglobulin as a clue to the diagnosis of thyrotoxicosis factitia. N Engl J Med. 1982;307:410–412.
270. Mariotti, S, Caturegli, P, Piccolo, P, et al. Antithyroid peroxidase autoantibodies in thyroid diseases. J Clin Endocrinol Metab. 1990;71:661–669.
271. McLachlan, SM, Rapoport, B. Thyroid peroxidase as an autoantigen. Thyroid. 2007;17:939–994.
272. McLachlan, SM, Rapoport, B. Why measure thyroglobulin autoantibodies rather than thyroid peroxidase autoantibodies? Thyroid. 2004;14:510–520.
273. Mariotti, S, Barbesino, G, Caturegli, P, et al. Assay of thyroglobulin in serum with thyroglobulin autoantibodies: an unobtainable goal? J Clin Endocrinol Metab. 1995;80:468–472.
274. Mariotti, S, Sansoni, P, Barbesino, G, et al. Thyroid and other organ-specific autoantibodies in healthy centenarians. Lancet. 1992;339:1506–1508.
275. Macchia, E, Concetti, R, Borgoni, F, et al. Assays of TSH-receptor antibodies in 576 patients with various thyroid disorders: Their incidence, significance and clinical usefulness. Autoimmunity. 1989;3:103–112.
276. Pittman, JA, Jr., Dailey, GE, 3d., Beschi, RJ. Changing normal values for thyroidal radioiodine uptake. N Engl J Med. 1969;280:1431–1434.
277. O’Hare, NJ, Murphy, D, Malone, JF. Thyroid dosimetry of adult European populations. Br J Radiol. 1998;71:535–543.
278. Martino, E, Bartalena, L, Mariotti, S, et al. Radioactive iodine thyroid uptake in patients with amiodarone-iodine–induced thyroid dysfunction. Acta Endocrinol (Copenh). 1988;119:167–173.
279. Gutekunst, R, Hafermann, W, Mansky, T, et al. Ultrasonography related to clinical and laboratory findings in lymphocytic thyroiditis. Acta Endocrinol (Copenh). 1989;121:129–135.
280. Vitti, P. Grey scale thyroid ultrasonography in the evaluation of patients with Graves’ disease. Eur J Endocrinol. 2000;142:22–24.
281. Vitti, P, Martino, E, Aghini-Lombardi, F, et al. Thyroid volume measurement by ultrasound in children as a tool for the assessment of mild iodine deficiency. J Clin Endocrinol Metab. 1994;79:600–603.
282. Bogazzi, F, Vitti, P. Could improved ultrasound and power Doppler replace thyroidal radioiodine uptake to assess thyroid disease? Nat Clin Pract Endocrinol Metab. 2008;4:70–71.
283. Cooper, DS. Antithyroid drugs. N Engl J Med. 1984;311:1353–1362.
284. Reinwein, D, Benker, G, Lazarus, JH, et al. A prospective randomized trial of antithyroid drug dose in Graves’ disease therapy. European Multicenter Study Group on Antithyroid Drug Treatment. J Clin Endocrinol Metab. 1993;76:1516–1521.
285. Hedley, AJ, Young, RE, Jones, SJ, et al. Antithyroid drugs in the treatment of hyperthyroidism of Graves’ disease: long-term follow-up of 434 patients. Scottish Automated Follow-Up Register Group. Clin Endocrinol (Oxf). 1989;31:209–218.
286. Schleusener, H, Schwander, J, Fischer, C, et al. Prospective multicentre study on the prediction of relapse after antithyroid drug treatment in patients with Graves’ disease [published erratum appears in Acta Endocrinol (Copenh) 1989 Aug;121(2):304]. Acta Endocrinol (Copenh). 1989;120:689–701.
287. Vitti, P, Rago, T, Chiovato, L, et al. Clinical features of patients with Graves’ disease undergoing remission after antithyroid drug treatment. Thyroid. 1997;7:369–375.
288. Kaguelidou, F, Alberti, C, Castanet, M, et al. Predictors of Autoimmune Hyperthyroidism Relapse in Children after Discontinuation of Antithyroid Drug Treatment. J Clin Endocrinol Metab. 2008 Jul 15.
289. Wartofsky, L. Low remission after therapy for Graves disease. Possible relation of dietary iodine with antithyroid therapy results. JAMA. 1973;226:1083–1088.
290. Solomon, BL, Evaul, JE, Burman, KD, et al. Remission rates with antithyroid drug therapy: continuing influence of iodine intake? Ann Intern Med. 1987;107:510–512.
291. Davies, TF, Yeo, PP, Evered, DC, et al. Value of thyroid-stimulating-antibody determinations in predicting short-term thyrotoxic relapse in Graves’ disease. Lancet. 1977;1:1181–1182.
292. Eshoj, O, Kvetny, J, Mogensen, EF, et al. Prediction of the course of Graves’ disease after medical antithyroid treatment. Acta Med Scand. 1985;217:225–228.
293. Nagataki, S. Prediction of relapse in Graves’ disease. Ann Acad Med Singapore. 1986;15:486–491.
294. Weetman, AP, Ratanachaiyavong, S, Middleton, GW, et al. Prediction of outcome in Graves’ disease after carbimazole treatment. Q J Med. 1986;59:409–419.
295. Wilson, R, McKillop, JH, Henderson, N, et al. The ability of the serum thyrotrophin receptor antibody (TRAb) index and HLA status to predict long-term remission of thyrotoxicosis following medical therapy for Graves’ disease. Clin Endocrinol (Oxf). 1986;25:151–156.
296. Talbot, JN, Duron, F, Feron, R, et al. Thyroglobulin, thyrotropin and thyrotropin binding inhibiting immunoglobulins assayed at the withdrawal of antithyroid drug therapy as predictors of relapse of Graves’ disease within one year. J Endocrinol Invest. 1989;12:589–595.
297. Ikenoue, H, Okamura, K, Sato, K, et al. Prediction of relapse in drug-treated Graves’ disease using thyroid stimulation indices. Acta Endocrinol (Copenh). 1991;125:643–650.
298. Lamberg, BA, Salmi, J, Wagar, G, et al. Spontaneous hypothyroidism after antithyroid treatment of hyperthyroid Graves’ disease. J Endocrinol Invest. 1981;4:399–402.
299. Feldt-Rasmussen, U, Schleusener, H, Carayon, P. Meta-analysis evaluation of the impact of thyrotropin receptor antibodies on long term remission after medical therapy of Graves’ disease. J Clin Endocrinol Metab. 1994;78:98–102.
300. Romaldini, JH, Werner, MC, Bromberg, N, et al. Adverse effects related to antithyroid drugs and their dose regimen. Exp Clin Endocrinol. 1991;97:261–264.
301. Davies, TF, Yeo, PP, Evered, DC, et al. Value of thyroid-stimulating-antibody determinations in predicting short-term thyrotoxic relapse in Graves’ disease. Lancet. 1977;1:1181–1182.
302. McGregor, AM, Petersen, MM, McLachlan, SM, et al. Carbimazole and the autoimmune response in Graves’ disease. N Engl J Med. 1980;303:302–304.
303. Marcocci, C, Chiovato, L, Mariotti, S, et al. Changes of circulating thyroid autoantibody levels during and after the therapy with methimazole in patients with Graves’ disease. J Endocrinol Invest. 1982;5:13–19.
304. Weetman, AP, McGregor, AP, Hall, R. Methimazole inhibits thyroid autoantibody production by an action on accessory cells. Clin Immunol Immunopathol. 1983;28:39–45.
305. Weiss, I, Davies, TF. Inhibition of immunoglobulin-secreting cells by antithyroid drugs. J Clin Endocrinol Metab. 1981;53:1223–1228.
306. Rennie, DP, McGregor, AM, Keats, D, et al. The influence of methimazole on thyroglobulin-induced autoimmune thyroiditis in the rat. Endocrinology. 1983;112:326–330.
307. Wenzel, KW, Lente, JR. Similar effects of thionamide drugs and perchlorate on thyroid-stimulating immunoglobulins in Graves’ disease: evidence against an immunosuppressive action of thionamide drugs. J Clin Endocrinol Metab. 1984;58:62–69.
308. Mariotti, S, Pinchera, A. Role of the immune system in the control of thyroid function. In: Greer MA, ed. The Thyroid Gland. New York: Raven; 1990:147–218.
309. Hashizume, K, Ichikawa, K, Sakurai, A, et al. Administration of thyroxine in treated Graves’ disease—effects on the level of antibodies to thyroid stimulating hormone receptors and on the risk of recurrence of hyperthyroidism. N Engl J Med. 1991;324:947–953.
310. Romaldini, JH, Bromberg, N, Werner, RS, et al. Comparison of effects of high and low dosage regimens of antithyroid drugs in the management of Graves’ hyperthyroidism. J Clin Endocrinol Metab. 1983;57:563–570.
311. Weetman, AP, Pickerill, AP, Watson, P, et al. Treatment of Graves’ disease with the block-replace regimen of antithyroid drugs: The effect of treatment duration and immunogenetic susceptibility on relapse. Q J Med. 1994;87:337–341.
312. Tamai, H, Hayaki, I, Kawai, K, et al. Lack of effect of thyroxine administration on elevated thyroid stimulating hormone receptor antibody levels in treated Graves’ disease patients. J Clin Endocrinol Metab. 1995;80:1481–1484.
313. Rittmaster, RS, Zwicker, H, Abbott, EC, et al. Effect of methimazole with or without exogenous l-thyroxine on serum concentrations of thyrotropin (TSH) receptor antibodies in patients with Graves’ disease. J Clin Endocrinol Metab. 1996;81:3283–3288.
314. Rizvi, A, Crapo, LM. Failure of thyroxine therapy for Graves disease (letter). Ann Intern Med. 1996;124:694.
315. Rittmaster, RS, Abbott, EC, Douglas, R, et al. Effect of methimazole, with or without l-thyroxine, on remission rates in Graves’ disease. J Clin Endocrinol Metab. 1998;83:814–818.
316. Emerson, CH, Anderson, AJ, Howard, WJ, et al. Serum thyroxine and triiodothyronine concentrations during iodide treatment of hyperthyroidism. J Clin Endocrinol Metab. 1975;40:33–36.
317. Marigold, JH, Morgan, AK, Earle, DJ, et al. Lugol's iodine: its effect on thyroid blood flow in patients with thyrotoxicosis. Br J Surg. 1985;72:45–47.
318. Chang, DC, Wheeler, MH, Woodcock, JP, et al. The effect of preoperative Lugol's iodine on thyroid blood flow in patients with Graves’ hyperthyroidism. Surgery. 1987;102:1055–1061.
319. Burgi, H, Wimpfheimer, C, Burger, A, et al. Changes of circulating thyroxine, triiodothyronine and reverse triiodothyronine after radiographic contrast agents. J Clin Endocrinol Metab. 1976;43:1203–1210.
320. Wu, SY, Chopra, IJ, Solomon, DH, et al. Changes in circulating iodothyronines in euthyroid and hyperthyroid subjects given ipodate (Oragrafin), an agent for oral cholecystography. J Clin Endocrinol Metab. 1978;46:691–697.
321. Bogazzi, F, Bartalena, L, Cosci, C, et al. Treatment of type II amiodarone-induced thyrotoxicosis by either iopanoic acid or glucocorticoids: a prospective, randomized study. J Clin Endocrinol Metab. 2003;88:1999–2002.
322. Robuschi, G, Manfredi, A, Salvi, M, et al. Effect of sodium ipodate and iodide on free T4 and free T3 concentrations in patients with Graves’ disease. J Endocrinol Invest. 1986;9:287–291.
323. Laurberg, P, Boye, N. Inhibitory effect of various radiographic contrast agents on secretion of thyroxine by the dog thyroid and on peripheral and thyroidal deiodination of thyroxine to tri-iodothyronine. J Endocrinol. 1987;112:387–390.
324. Wu, SY, Shyh, TP, Chopra, IJ, et al. Comparison of sodium ipodate (Oragrafin) and propylthiouracil in early treatment of hyperthyroidism. J Clin Endocrinol Metab. 1982;54:630–634.
325. Shen, DC, Wu, SY, Chopra, IJ, et al. Long-term treatment of Graves’ hyperthyroidism with sodium ipodate. J Clin Endocrinol Metab. 1985;61:723–727.
326. Martino, E, Balzano, S, Bartalena, L, et al. Therapy of Graves’ disease with sodium ipodate is associated with a high recurrence rate of hyperthyroidism. J Endocrinol Invest. 1991;14:847–851.
327. DeGroot, LJ, Buhler, U. Effect of perchlorate and methimazole on iodine metabolism. Acta Endocrinol (Copenh). 1971;68:696–706.
328. Barzilai, D, Sheinfeld, M. Fatal complications following use of potassium perchlorate in thyrotoxicosis. Report of two cases and a review of the literature. Isr J Med Sci. 1966;2:453–456.
329. Bartalena, L, Brogioni, S, Grasso, L, et al. Treatment of amiodarone-induced thyrotoxicosis, a difficult challenge: results of a prospective study. J Clin Endocrinol Metab. 1996;81:2930–2933.
330. Bogazzi, F, Bartalena, L, Campomori, A, et al. Treatment with lithium prevents serum thyroid hormone increase after thionamide withdrawal and radioiodine therapy in patients with Graves’ disease. J Clin Endocrinol Metab. 2002;87:4490–4495.
331. Bogazzi, F, Martino, E, Bartalena, L. Antithyroid drug treatment prior to radioiodine therapy for Graves’ disease: yes or no? J Endocrinol Invest. 2003;26:174–176.
332. Henderson, JM, Portmann, L, Van Melle, G, et al. Propranolol as an adjunct therapy for hyperthyroid tremor. Eur Neurol. 1997;37:182–185.
333. Wiersinga, WM. Propranolol and thyroid hormone metabolism. Thyroid. 1991;1:273–277.
334. Dobyns, BM, Vickery, AL, Maloof, F, et al. Functional and histologic effects of therapeutic doses of radioiodine therapy for hyperthyroidism. J Clin Endcrinol Metab. 1953;13:548.
335. Beierwaltes, WH. The treatment of hyperthyroidism with iodine-131. Semin Nucl Med. 1978;8:95–103.
336. Franklyn, JA, Daykin, J, Drolc, Z, et al. Long-term follow-up of treatment of thyrotoxicosis by three different methods. Clin Endocrinol (Oxf). 1991;34:71–76.
337. Bartalena, L, Marcocci, C, Bogazzi, F, et al. Relation between therapy for hyperthyroidism and the course of Graves’ ophthalmopathy. N Engl J Med. 1998;338:73–78.
338. Perros, P, Kendall-Taylor, P, Neoh, C, et al. A prospective study of the effects of radioiodine therapy for hyperthyroidism in patients with minimally active Graves’ ophthalmopathy. J Clin Endocrinol Metab. 2005;90:5321–5323.
339. Burch, HB, Solomon, BL, Wartofsky, L, et al. Discontinuing antithyroid drug therapy before ablation with radioiodine in Graves’ disease. Ann Intern Med. 1994;121:553–559.
340. Block, MA, Miller, MJ, Horn, RC, Jr. Carcinoma of the thyroid after external radiation to the neck in adults. Am J Surg. 1969;118:764–769.
341. Ron, E, Saftlas, AF. Head and neck radiation carcinogenesis: epidemiologic evidence. Otolaryngol Head Neck Surg. 1996;115:403–408.
342. Hall, P, Holm, LE. Late consequences of radioiodine for diagnosis and therapy in Sweden. Thyroid. 1997;7:205–208.
343. Ron, E, Doody, MM, Becker, DV, et al. Cancer mortality following treatment for adult hyperthyroidism. Cooperative Thyrotoxicosis Therapy Follow-up Study Group. JAMA. 1998;280:347–355.
344. Graham, GD, Burman, KD. Radioiodine treatment of Graves’ disease. An assessment of its potential risks. Ann Intern Med. 1986;105:900–905.
345. Federico, G, Boni, G, Fabiani, B, et al. No evidence of chromosome damage in children and adolescents with differentiated thyroid carcinoma after receiving 131I radiometabolic therapy, as evaluated by micronucleus assay and microarray analysis. Eur J Nucl Med Mol Imaging.. 2008.
346. Hennemann, G, Krenning, EP, Sankaranarayanan, K. Place of radioactive iodine in treatment of thyrotoxicosis. Lancet. 1986;1:1369–1372.
347. Franklyn, JA, Sheppard, MC, Maisonneuve, P. Thyroid function and mortality in patients treated for hyperthyroidism. JAMA. 2005;294:71–80.
348. Baverstock, K, Egloff, B, Pinchera, A, et al. Thyroid cancer after Chernobyl. Nature. 1992;359:21–22.
349. Pacini, F, Vorontsova, T, Demidchik, EP, et al. Post-Chernobyl thyroid carcinoma in Belarus children and adolescents: comparison with naturally occurring thyroid carcinoma in Italy and France. J Clin Endocrinol Metab. 1997;82:3563–3569.
350. Dobyns, BM, Sheline, GE, Workman, JB, et al. Malignant and benign neoplasms of the thyroid in patients treated for hyperthyroidism: a report of the Cooperative Thyrotoxicosis Therapy Follow-Up Study. J Clin Endocrinol Metab. 1974;38:976–998.
351. Rivkees, SA, Dinauer, C. An optimal treatment for pediatric Graves’ disease is radioiodine. J Clin Endocrinol Metab. 2007;92:797–800.
352. Miccoli, P, Vitti, P, Rago, T, et al. Surgical treatment of Graves’ disease: subtotal or total thyroidectomy? Surgery. 1996;120:1020–1025.
353. Kasuga, Y, Sugenoya, A, Kobayashi, S, et al. Clinical evaluation of the response to surgical treatment of Graves’ disease. Surg Gynecol Obstet. 1990;170:327–330.
354. Menconi, F, Marinò, M, Pinchera, A, et al. Effects of total thyroid ablation versus near-total thyroidectomy alone on mild to moderate Graves’ orbitopathy treated with intravenous glucocorticoids. J Clin Endocrinol Metab. 2007;92:1653–1658.
355. Baeza, A, Aguayo, J, Barria, M, et al. Rapid preoperative preparation in hyperthyroidism. Clin Endocrinol (Oxf). 1991;35:439–442.
356. Tomaski, SM, Mahoney, EM, Burgess, LP, et al. Sodium ipodate (Oragrafin) in the preoperative preparation of Graves’ hyperthyroidism. Laryngoscope. 1997;107:1066–1070.
357. Max, MH, Scherm, M, Bland, KI. Early and late complications after thyroid operations. South Med J. 1983;76:977–980.
358. Kasemsuwan, L, Nubthuenetr, S. Recurrent laryngeal nerve paralysis: a complication of thyroidectomy. J Otolaryngol. 1997;26:365–367.
359. Pattou, F, Combemale, F, Fabre, S, et al. Hypocalcemia following thyroid surgery: incidence and prediction of outcome. World J Surg. 1998;22:718–724.
360. Glinoer, D, Hesch, D, Lagasse, R, et al. The management of hyperthyroidism due to Graves’ disease in Europe in 1986. Results of an international survey. Acta Endocrinol Suppl. 1987;285:3–23.
361. Wartofsky, L, Glinoer, D, Solomon, B, et al. Differences and similarities in the treatment of diffuse goiter in Europe and the United States. Exp Clin Endocrinol. 1991;97:243–251.
362. Freitas, JE, Swanson, DP, Gross, MD, et al. Iodine-131: optimal therapy for hyperthyroidism in children and adolescents? J Nucl Med. 1979;20:847–850.
363. Rivkees, SA, Sklar, C, Freemark, M. Clinical review 99: the management of Graves’ disease in children, with special emphasis on radioiodine treatment. J Clin Endocrinol Metab. 1998;83:3767–3776.
364. Waldhausen, JH. Controversies related to the medical and surgical management of hyperthyroidism in children. Semin Pediatr Surg. 1997;6:121–127.
365. Witte, J, Goretzki, PE, Roher, HD. Surgery for Graves’ disease in childhood and adolescence. Exp Clin Endocrinol Diabetes. 1997;105:58–60.
366. Lazarus, JH. Guidelines for the use of radioiodine in the management of hyperthyroidism: a summary. Prepared by the Radioiodine Audit Subcommittee of the Royal College of Physicians Committee on Diabetes and Endocrinology, and the Research Unit of the Royal College of Physicians. J R Coll Physicians Lond. 1995;29:464–469.
367. Weetman, AP, Wiersinga, WM. Current management of thyroid-associated ophthalmopathy in Europe. Results of an international survey. Clin Endocrinol (Oxf). 1998;49:21–28.
368. Chiovato, L, Latrofa, F, Braverman, LE, et al. Disappearance of humoral thyroid autoimmunity after complete removal of thyroid antigens. Ann Int Med. 2003;139:346–351.
369. Abalovich, M, Amino, N, Barbour, LA, et al. Management of thyroid dysfunction during pregnancy and postpartum: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2007;92(8 Suppl):S1–47.
370. Chattaway, JM, Klepser, TB. Propylthiouracil versus methimazole in treatment of Graves’ disease during pregnancy. Ann Pharmacother. 2007;41:1018–1022.
371. Marchant, B, Brownlie, BE, Hart, DM, et al. The placental transfer of propylthiouracil, methimazole and carbimazole. J Clin Endocrinol Metab. 1977;45:1187–1193.
372. Cheron, RG, Kaplan, MM, Larsen, PR, et al. Neonatal thyroid function after propylthiouracil therapy for maternal Graves’ disease. N Engl J Med. 1981;304:525–528.
373. Ribuffo, D, Costantini, M, Gullo, P, et al. Aplasia cutis congenita of the scalp, the skull, and the dura. Scand J Plast Reconstr Surg Hand Surg. 2003;37:176–180.
374. Santini, F, Chiovato, L, Ghirri, P, et al. Serum iodothyronines in the human fetus and the newborn: evidence for an important role of placenta in fetal thyroid hormone homeostasis. J Clin Endocrinol Metab. 1999;84:493–498.
375. Lamberg, BA, Ikonen, E, Osterlund, K, et al. Antithyroid treatment of maternal hyperthyroidism during lactation. Clin Endocrinol (Oxf). 1984;21:81–87.
376. Zakarija, M, McKenzie, JM. Pregnancy-associated changes in the thyroid-stimulating antibody of Graves’ disease and the relationship to neonatal hyperthyroidism. J Clin Endocrinol Metab. 1983;57:1036–1040.
377. Tamaki, H, Amino, N, Takeoka, K, et al. Prediction of later development of thyrotoxicosis or central hypothyroidism from the cord serum thyroid-stimulating hormone level in neonates born to mothers with Graves’ disease. J Pediatr. 1989;115:318–321.
378. Karpman, BA, Rapoport, B, Filetti, S, et al. Treatment of neonatal hyperthyroidism due to Graves’ disease with sodium ipodate. J Clin Endocrinol Metab. 1987;64:119–123.
379. Trivalle, C, Doucet, J, Chassagne, P, et al. Differences in the signs and symptoms of hyperthyroidism in older and younger patients. J Am Geriatr Soc. 1996;44:50–53.
380. Martin, FI, Deam, DR. Hyperthyroidism in elderly hospitalised patients. Clinical features and treatment outcomes. Med J Aust. 1996;164:200–203.
381. Perros, P, Crombie, AL, Matthews, JN, et al. Age and gender influence the severity of thyroid-associated ophthalmopathy: a study of 101 patients attending a combined thyroid-eye clinic. Clin Endocrinol (Oxf). 1993;38:367–372.
382. Kriss, JP, Pleshakov, V, Rosenblum, A, et al. Therapy with occlusive dressings of pretibial myxedema with fluocinolone acetonide. J Clin Endocrinol Metab. 1967;27:595–604.
383. Lang, PG, Sisson, JC, Lynch, PJ. Intralesional triamcinolone therapy for pretibial myxedema. Arch Dermatol. 1975;111:197–202.
384. Derrick, EK, Tanner, B, Price, ML. Successful surgical treatment of severe pretibial myxoedema. Br J Dermatol. 1995;133:317–318.
385. Pingsmann, A, Ockenfels, HM, Patsalis, T. Surgical excision of pseudotumorous pretibial myxedema. Foot Ankle Int. 1996;17:107–110.
386. Wartofsky, L. Treatment options for hyperthyroidism. Hosp Pract. 1996;31:69–73.